Table of Contents
Overview
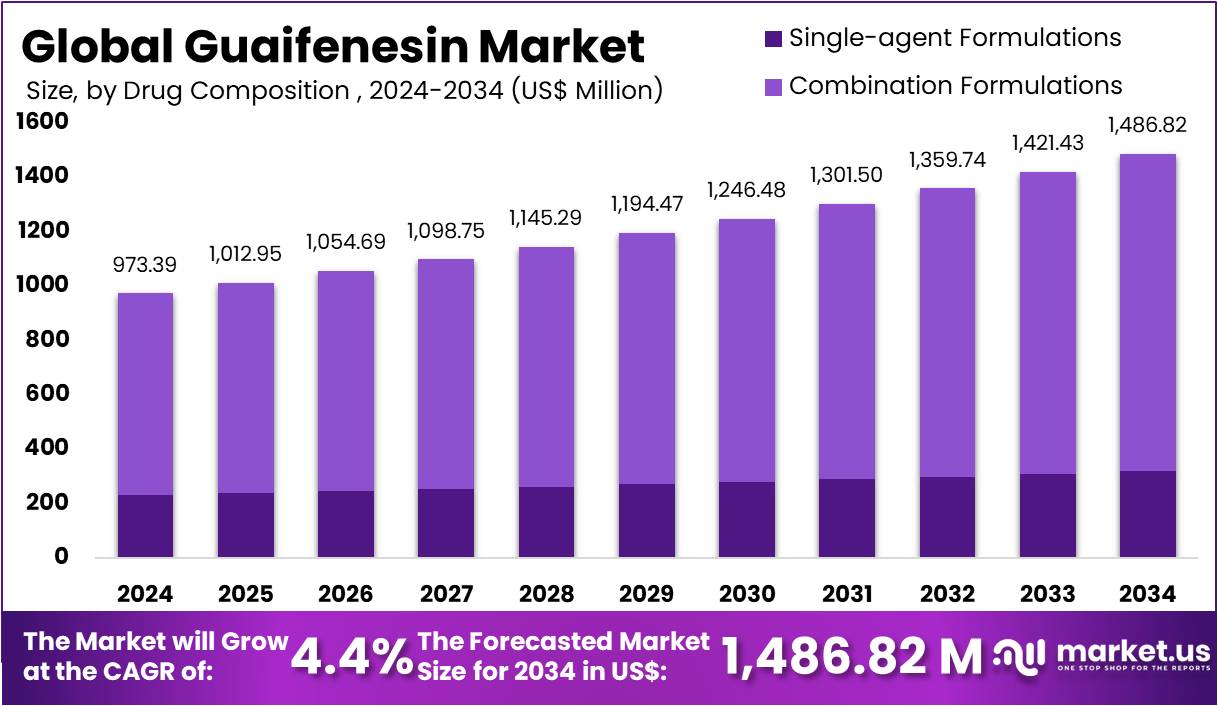
New York, NY – May 05, 2026 – The Global Guaifenesin Market size is expected to be worth around US$ 1,486.82 Million by 2034 from US$ 973.39 Million in 2024, growing at a CAGR of 4.4% during the forecast period 2025 to 2034.
Guaifenesin is a widely used pharmaceutical ingredient classified as an expectorant, primarily indicated for the symptomatic relief of chest congestion associated with respiratory conditions such as acute bronchitis, common cold, and upper respiratory tract infections. Its mechanism of action involves increasing the hydration of respiratory tract secretions, thereby reducing mucus viscosity and facilitating easier expectoration.
The compound is commonly formulated in oral dosage forms, including tablets, extended-release capsules, and syrups, often in combination with other active ingredients such as decongestants or antitussives. The global demand for guaifenesin-based products has been supported by the rising incidence of respiratory illnesses, seasonal infections, and increasing consumer preference for over-the-counter (OTC) medications.
From a safety perspective, guaifenesin is generally well tolerated when administered within recommended dosage limits. Mild adverse effects, including gastrointestinal discomfort, nausea, or dizziness, have been reported in a limited number of cases. Regulatory authorities such as the U.S. Food and Drug Administration have approved its use in various OTC formulations, reinforcing its established safety and efficacy profile.
Market growth is being driven by expanding healthcare access, growing awareness of self-medication, and continuous product innovation. The outlook for guaifenesin remains cautiously positive, supported by sustained demand in both developed and emerging pharmaceutical markets.
Key Takeaways
- In 2024, the Guaifenesin market generated revenue of approximately US$ 39 million and is projected to expand at a CAGR of 4.4%, reaching US$ 1,486.82 million by 2034.
- Combination formulations dominated the market by drug composition, accounting for a 76.1% share in 2024.
- By purity level, products with purity greater than 99% led the segment, generating revenue of US$ 612.99 million in 2024.
- Based on formulation type, the market is categorized into tablets and capsules, syrups/solutions, granules/extended-release, and others. Among these, tablets and capsules held the largest share of 35% in 2024.
- In terms of application, the cough suppressant segment emerged as the leading category, capturing 45.2% of the market share in 2024.
- By distribution channel, retail pharmacies accounted for the highest share, contributing 58.2% in 2024.
- Regionally, North America led the global market, securing a share of 34.1% in 2024.
Regional Analysis
North America accounted for the largest share of the Guaifenesin market, contributing 34.1% of total revenue in 2024. This regional dominance is expected to persist, supported by the sustained prevalence of respiratory conditions across the United States and Canada.
Epidemiological data indicate consistent incidence rates of respiratory syncytial virus (RSV)–associated acute respiratory illness between October 2022 and April 2024, with positivity rates ranging from 5.5% to 5.8% across observed seasons. The continued burden of common respiratory infections, including cold and influenza, is driving demand for effective symptomatic treatments.
Additionally, the widespread availability of guaifenesin as an over-the-counter medication enhances accessibility, contributing to its high utilization. Prescription data further reinforce market penetration, with more than 400,000 prescriptions recorded in the United States in 2022.
The Asia Pacific region is projected to register the fastest growth rate over the forecast period. Market expansion is being driven by increasing incidence of respiratory infections, as reported by the World Health Organization in early 2025.
Rising air pollution levels have also been identified as a critical factor, contributing to higher respiratory disease burden. Limited compliance with WHO air quality standards across several countries is expected to further accelerate demand for expectorant therapies.
Emerging Trends
- Rising Focus on Chronic Respiratory Care: The application of guaifenesin has been expanding beyond short-term respiratory infections toward long-term disease management. Increasing adoption has been observed among patients with chronic conditions such as chronic bronchitis and COPD. Its mucolytic properties support the thinning and clearance of persistent mucus, thereby improving airway function. Ongoing clinical evaluations are assessing its sustained use and impact on quality of life. This shift indicates a broader positioning of guaifenesin as a supportive therapy in chronic respiratory care rather than solely an acute treatment option.
- Increasing Adoption of Extended-Release Formulations: Extended-release guaifenesin formulations are gaining significant traction in the market. These formulations enable gradual drug release, ensuring prolonged therapeutic action and reducing dosing frequency. Improved patient adherence has been noted, particularly among geriatric populations and individuals requiring continuous symptom management. The convenience associated with once- or twice-daily dosing aligns with evolving consumer preferences for simplified treatment regimens. Pharmaceutical manufacturers are increasingly investing in such formulations to enhance patient compliance and product differentiation.
- Variable Efficacy in Acute Respiratory Conditions: Clinical outcomes associated with guaifenesin use in short-term conditions, such as common cold and influenza, remain inconsistent. While certain patient groups report symptomatic relief in mucus clearance and cough productivity, others demonstrate limited response. This variability has prompted further research aimed at refining clinical evaluation methodologies and improving efficacy measurement parameters. Enhanced understanding of its performance in acute conditions is expected to support optimized formulation development and more targeted therapeutic recommendations.
- Growth of Combination Therapy Products: The integration of guaifenesin into multi-ingredient formulations continues to expand within the over-the-counter (OTC) segment. These combination products typically include cough suppressants, antihistamines, and decongestants, offering comprehensive symptom relief. Market demand for convenient, all-in-one solutions has accelerated product innovation in this category. The approach reduces treatment complexity and enhances user convenience, positioning combination therapies as a dominant trend in the cough and cold therapeutics market.
Use Cases
- Management of Chest Congestion in Cold and Flu: Guaifenesin is widely utilized in the treatment of respiratory infections such as colds and influenza, where mucus accumulation leads to chest congestion. Its mechanism facilitates the loosening and thinning of mucus, promoting effective expectoration. This contributes to improved respiratory comfort and faster clearance of airway blockages. Available in multiple dosage forms, it remains a standard component of symptomatic treatment protocols.
- Supportive Therapy in Chronic Respiratory Disorders: In chronic respiratory conditions, including asthma, chronic bronchitis, and COPD, guaifenesin is used as an adjunct therapy to manage mucus-related complications. Regular administration helps maintain airway clearance and reduces the frequency of productive coughing episodes. Although it does not address the underlying pathology, its role in symptom control enhances patient comfort and supports long-term disease management strategies.
- Inclusion in Multi-Symptom OTC Formulations: Guaifenesin is a core ingredient in numerous OTC multi-symptom medications designed for cold and flu relief. These formulations combine multiple active agents to address cough, nasal congestion, fever, and other associated symptoms simultaneously. The convenience of single-product usage has contributed to strong consumer preference and sustained demand within this segment.
- Use in Prescription-Based Combination Therapies: In cases of severe or persistent cough, guaifenesin is incorporated into prescription medications alongside stronger agents such as codeine. This combination provides dual therapeutic action—mucus clearance and cough suppression. Such treatments are typically recommended under medical supervision for conditions including post-operative recovery, severe infections, or chronic respiratory disorders. The inclusion of guaifenesin enhances overall treatment efficacy in these advanced therapeutic interventions.
Frequently Asked Questions on Guaifenesin
- What are the primary uses of Guaifenesin?
Guaifenesin is primarily indicated for managing symptoms associated with respiratory conditions such as the common cold, bronchitis, and upper respiratory tract infections. It is widely used to reduce mucus buildup and improve airway clearance in patients experiencing productive cough. - Is Guaifenesin safe for regular use?
Guaifenesin is generally considered safe when used within recommended dosage guidelines. Mild side effects such as nausea, dizziness, or gastrointestinal discomfort may occur in some individuals. Regulatory approvals have established its safety profile for over-the-counter and prescription-based use. - What factors are driving the growth of the Guaifenesin market?
The growth of the guaifenesin market is driven by rising respiratory disease prevalence, increasing self-medication practices, and widespread availability of over-the-counter formulations. Expanding healthcare access and growing awareness regarding cough management further support sustained market demand. - Which regions are leading in the Guaifenesin market?
North America currently leads the guaifenesin market due to high consumption of respiratory medications and strong healthcare infrastructure. However, Asia Pacific is expected to witness the fastest growth, driven by increasing pollution levels and rising incidence of respiratory infections. - What are the key product segments in the Guaifenesin market?
Key product segments include tablets and capsules, syrups, and extended-release formulations. Among these, tablets and capsules account for a significant share due to ease of administration, longer shelf life, and widespread consumer preference across various demographic groups. - How is Guaifenesin distributed in the market?
Guaifenesin products are primarily distributed through retail pharmacies, hospital pharmacies, and online platforms. Retail pharmacies dominate the distribution channel due to their accessibility and the over-the-counter availability of guaifenesin-based formulations in most regions.
Conclusion
The guaifenesin market demonstrates stable growth, supported by rising respiratory disease prevalence and increasing reliance on over-the-counter therapies. Its established safety profile, broad application in both acute and chronic conditions, and expanding use in combination formulations reinforce its market relevance.
North America maintains a leading position, while Asia Pacific is expected to witness accelerated growth due to environmental and epidemiological factors. Continued innovation, particularly in extended-release formats and multi-symptom products, is expected to enhance patient compliance and product differentiation.
Overall, the market outlook remains cautiously positive, driven by sustained demand and evolving therapeutic applications.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



