Table of Contents
Overview
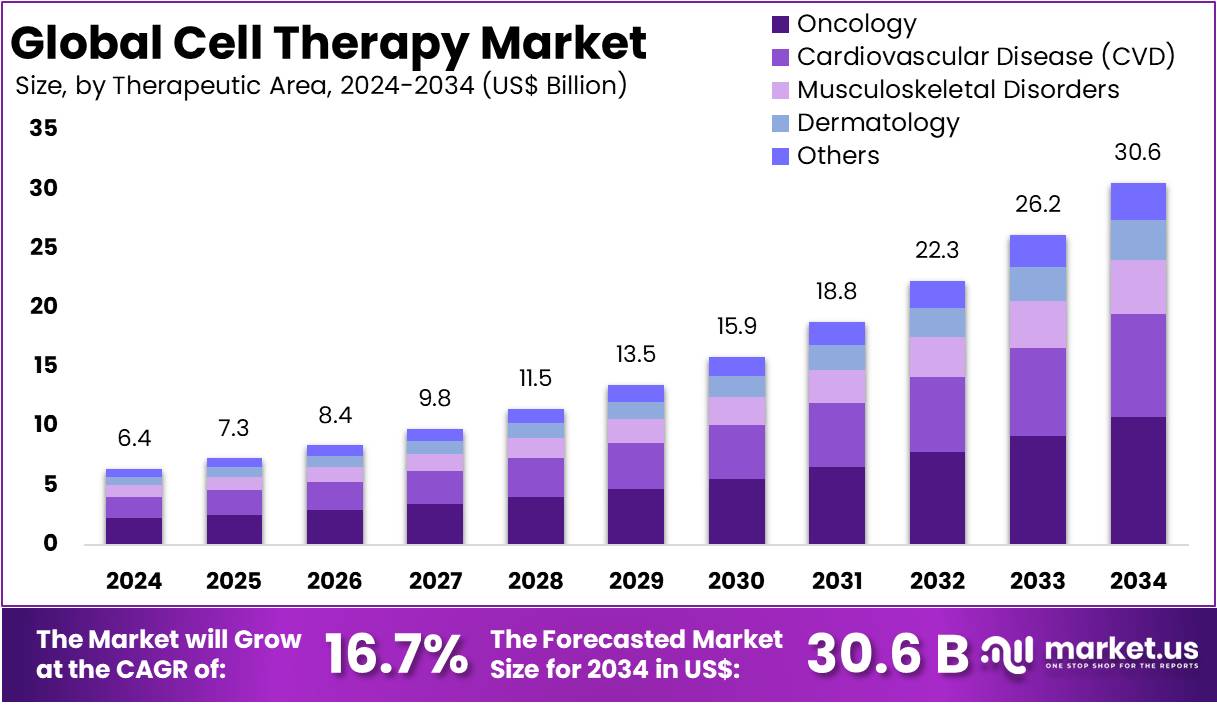
New York, NY – April 29, 2026 – The Global Cell Therapy Market size is expected to be worth around US$ 30.6 Billion by 2034, from US$ 6.4 Billion in 2024, growing at a CAGR of 16.7% during the forecast period from 2025 to 2034. North America held a dominant market position, capturing more than a 53.1% share and holds US$ 3.3 Billion market value for the year.
Cell therapy is an advanced medical approach in which living cells are administered to patients to treat, manage, or potentially cure a wide range of diseases. These therapies utilize viable cells, often derived from the patient (autologous) or a donor (allogeneic), to repair or replace damaged tissues, restore normal function, or modulate immune responses. The approach has gained significant attention due to its potential to address conditions that are difficult to treat with conventional pharmaceuticals.
The scope of cell therapy includes applications in oncology, regenerative medicine, and autoimmune disorders. In cancer treatment, engineered cell therapies such as CAR-T cells are designed to recognize and eliminate malignant cells with high specificity. In regenerative medicine, stem cell-based therapies are being developed to repair tissues affected by conditions such as cardiovascular diseases, neurological disorders, and musculoskeletal injuries.
Technological advancements, including gene editing, cell expansion techniques, and improved manufacturing processes, have enhanced the safety, efficacy, and scalability of cell therapies. Regulatory support and increasing investment in biopharmaceutical research have further accelerated clinical development and commercialization.
Overall, cell therapy represents a transformative shift in modern medicine, offering targeted, personalized treatment solutions. Its continued evolution is expected to significantly improve patient outcomes and reshape the future landscape of healthcare delivery.
Key Takeaways
- The global cell therapy market was valued at USD 6.4 billion in 2024 and is projected to reach USD 30.6 billion by 2034, expanding at a CAGR of 16.7%.
- Autologous therapies dominated the market in 2024, accounting for 90.0% of the total revenue share.
- The oncology segment emerged as the leading application area, contributing 38.2% of global revenue.
- North America held the dominant regional position, capturing more than 53.1% of the overall market share.
Regional Analysis
The North America cell therapy market is witnessing strong expansion, supported by continuous advancements in medical research, a rising burden of chronic diseases, and favorable regulatory frameworks. The United States represents the largest contributor, while Canada also plays a significant role in regional growth. Market development is driven by high healthcare expenditure, supportive government initiatives, and the presence of well-established pharmaceutical and biotechnology companies.
The United States remains a global hub for biopharmaceutical innovation, significantly influencing the development and commercialization of cell therapies. Oncology-focused applications dominate, with substantial investments directed toward advanced solutions such as CAR-T cell therapies, which have transformed cancer treatment approaches.
Moreover, increasing focus on regenerative medicine for cardiovascular and neurological conditions is accelerating market adoption. The growing incidence of cancer continues to strengthen demand for cell-based therapies. According to the Centers for Disease Control and Prevention, approximately 1,603,844 new cancer cases were reported in the United States in 2020.
Emerging Trends
- Shift Toward Distributed Manufacturing Models: Cell therapy production is increasingly transitioning to decentralized manufacturing frameworks. Smaller, regionally located facilities are being utilized to produce therapies closer to patient populations, which reduces transportation complexity, minimizes logistical risks, and enhances treatment accessibility. Strategic collaborations between regulatory bodies and healthcare systems are supporting the development of reliable manufacturing networks for both routine and emergency applications.
- Adoption of Closed and Automated Bioreactor Systems: There is a growing implementation of closed-system bioreactors designed to automate key manufacturing stages under sterile conditions. These systems enable precise regulation of process variables such as fluid flow, shear stress, and environmental conditions, thereby reducing contamination risks and improving reproducibility and consistency of final products.
- Expansion of Regulatory Frameworks with Lifecycle Emphasis: Regulatory authorities have introduced updated guidance to address complexities in advanced cell therapy manufacturing. Recent draft guidelines on comparability and genome editing emphasize a lifecycle-based regulatory approach, requiring continuous monitoring and control of process modifications to maintain product quality and safety.
- Integration of Artificial Intelligence and Advanced Analytics: The use of artificial intelligence and machine learning is expanding within cell therapy manufacturing. These technologies are being applied for real-time process monitoring, anomaly detection, and optimization. Since 2016, a significant number of investigational submissions have incorporated AI/ML tools, indicating increasing regulatory confidence in data-driven manufacturing strategies.
Use Cases
- Autologous CAR-T Cell Therapies: Autologous CAR-T therapies involve the modification of a patient’s own T cells to target cancer cells, particularly in hematologic malignancies. The manufacturing process includes leukapheresis, genetic engineering, and ex vivo expansion. Multiple approved therapies have demonstrated strong clinical outcomes, with complete response rates reaching up to approximately 67% in certain lymphoma indications.
- Regenerative Medicine Advanced Therapies (RMAT): Therapies granted RMAT designation are targeting complex and rare conditions, including metastatic cancers and immune deficiencies. Recent product approvals rely on scalable manufacturing capabilities, including high-yield vector production and controlled cell culture processes, enabling the delivery of large, therapeutically effective doses with consistent quality.
Frequently Asked Questions on Cell Therapy
- How does cell therapy work?
Cell therapy works by introducing functional or engineered cells that can restore biological activity or stimulate regeneration. These cells may differentiate into required cell types or release bioactive molecules that promote tissue repair and immune system modulation. - What are the major types of cell therapy?
The major types include autologous therapies, using a patient’s own cells, and allogeneic therapies, derived from donors. Additionally, stem cell therapy and immune cell therapies, such as CAR-T cells, are prominent categories driving clinical and commercial adoption. - What diseases can be treated using cell therapy?
Cell therapy has shown effectiveness in treating cancers, cardiovascular diseases, neurodegenerative disorders, and autoimmune conditions. It is also being explored for regenerative applications in orthopedics and wound healing, reflecting a broad and expanding therapeutic scope. - What factors are driving the growth of the cell therapy market?
Market growth is driven by increasing prevalence of chronic diseases, advancements in biotechnology, and rising investment in regenerative medicine. Favorable regulatory support and growing clinical success rates are further strengthening adoption across developed and emerging markets. - Who are the major players in the cell therapy market?
The market is characterized by the presence of biotechnology firms, pharmaceutical companies, and research institutions. Strategic collaborations, mergers, and acquisitions are commonly observed, aimed at enhancing technological capabilities and expanding product pipelines globally. - What is the future outlook of the cell therapy market?
The market is expected to witness robust growth, driven by continuous innovation and expanding clinical applications. Increasing approvals and technological advancements in gene editing and cell engineering are anticipated to significantly enhance therapeutic efficacy and market penetration. - What role does innovation play in the cell therapy market?
Innovation plays a critical role, particularly in areas such as gene editing, automation, and scalable manufacturing. Continuous research and technological advancements are improving treatment outcomes, reducing costs, and enabling broader accessibility across diverse patient populations.
Conclusion
Cell therapy is positioned as a transformative segment within the global biopharmaceutical landscape, supported by strong clinical efficacy and expanding therapeutic applications. Market growth is being driven by rising chronic disease prevalence, advancements in gene editing and manufacturing technologies, and increasing regulatory support.
Dominance of autologous therapies and oncology applications highlights current commercialization trends, while regenerative medicine presents future growth opportunities. North America continues to lead due to robust infrastructure and investment.
Emerging innovations, including AI integration and decentralized manufacturing, are expected to enhance scalability, efficiency, and accessibility, reinforcing cell therapy’s long-term potential to reshape modern treatment paradigms and improve patient outcomes.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



