Table of Contents
Overview
New York, NY – Feb 27, 2026 – The Global Urokinase for Injection Market Size is expected to be worth around US$ 199.0 Million by 2033 from US$ 141.1 Million in 2023, growing at a CAGR of 3.5% during the forecast period 2024 to 2033.
Urokinase for Injection is a thrombolytic agent formulated to support the management of thromboembolic disorders. It is a sterile, lyophilized preparation containing urokinase, an enzyme that facilitates the conversion of plasminogen to plasmin, thereby promoting the degradation of fibrin clots. The product is intended for reconstitution with a suitable diluent prior to intravenous administration under controlled clinical settings.
The basic formulation typically comprises highly purified urokinase as the active pharmaceutical ingredient (API), along with stabilizing excipients such as human serum albumin or other approved protein stabilizers to maintain enzymatic activity and structural integrity during storage. Buffering agents may be included to ensure optimal pH stability, while the lyophilization process enhances product shelf life and ensures consistent potency.
Stringent manufacturing standards are applied throughout the production process to ensure batch-to-batch consistency, sterility, and compliance with regulatory quality requirements. The formulation is designed to deliver rapid onset of action, supporting effective thrombolysis in indicated clinical scenarios.
Growing demand for advanced thrombolytic therapies has contributed to sustained interest in urokinase-based formulations across hospital and specialty care settings. Continued focus on quality assurance, cold-chain logistics, and regulatory compliance remains essential to maintaining product reliability and therapeutic performance in global markets.
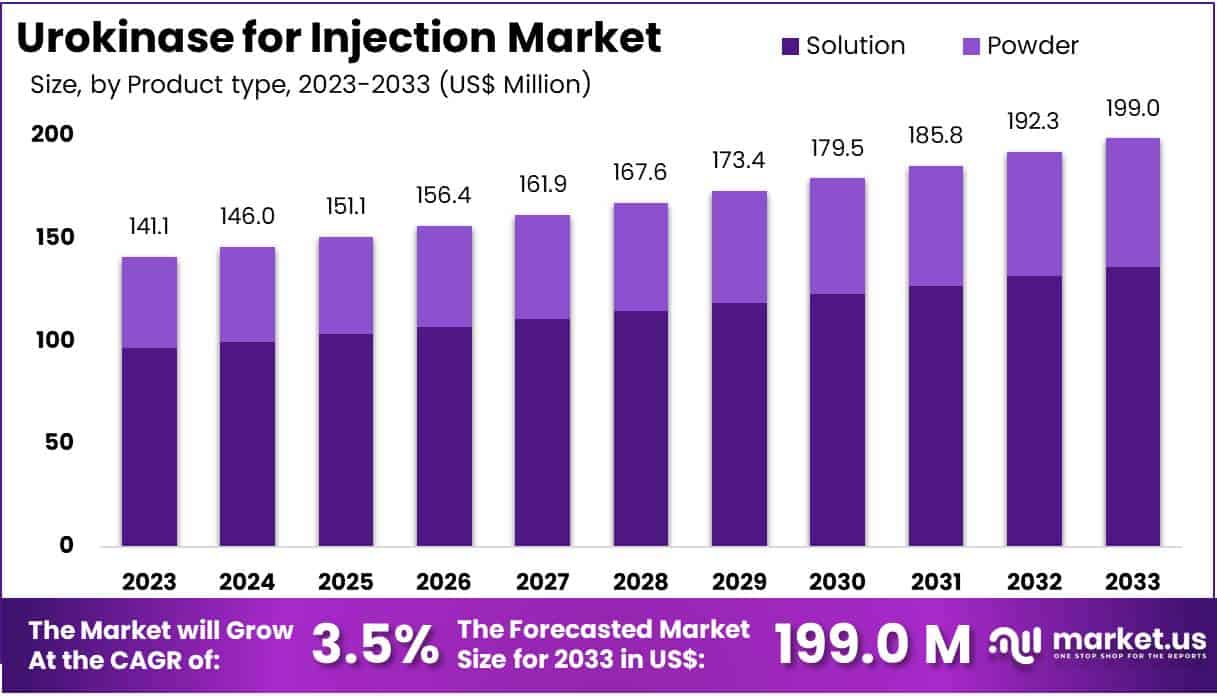
Key Takeaways
- In 2023, the urokinase for injection market generated revenue of US$ 141.1 million, with a CAGR of 3.5%, projected to reach US$ 199.0 million by 2033.
- By product type, the market is segmented into powder and solution, with solution leading at 68.5% market share in 2023.
- By application, the market includes hospitals, clinics, and others, with hospitals accounting for a dominant 64.3% share.
- North America led the market in 2023, holding a 40.2% share.
Regional Analysis
North America led the Urokinase for Injection market in 2023, accounting for 40.2% of total revenue. Market dominance was driven by the high incidence of thromboembolic disorders and sustained demand for effective clot-dissolving therapies. Cardiovascular diseases and pulmonary embolism remain major contributors to morbidity.
According to the Centers for Disease Control and Prevention, venous thromboembolism (VTE) affects nearly 900,000 individuals annually in the United States, reinforcing the need for advanced thrombolytic agents such as urokinase. An aging population and improved diagnostic capabilities have further supported treatment adoption across hospital settings.
Asia Pacific is projected to register the fastest CAGR during the forecast period. Growth is supported by rising cardiovascular disease prevalence, expanding healthcare infrastructure, and improved access to modern therapies. In Japan, 28.9% of the population was aged 65 or older in 2022, contributing to higher clot-related risk. Government healthcare initiatives and increasing local pharmaceutical production are expected to strengthen regional market expansion.
Emerging Trends
- Growing Use in Cancer Research: Urokinase is being explored to target cancer cells expressing urokinase-type plasminogen activator receptors (uPAR), aiming for precise tumor destruction while minimizing side effects of conventional therapies.
- Development of Safer Formulations: Manufacturers are improving urokinase formulations to enhance stability, reduce side effects, and simplify handling, enabling safer and more efficient use in hospitals and emergency medical settings.
- Expanded Role in Tuberculosis-Related Conditions: Urokinase is tested for pleural effusions caused by tuberculosis, helping break down fluid, improve breathing, and reduce surgical needs, especially in areas with limited medical access.
- Focus on Affordable Thrombolytic Therapy: Urokinase remains a cost-effective thrombolytic option compared with alternatives, supporting treatment accessibility in developing countries and public healthcare systems, addressing growing patient demand for affordable therapies.
Use Cases
- Treatment of Pulmonary Embolism (PE): Urokinase dissolves clots in pulmonary embolism, restoring blood flow, reducing cardiac and lung strain, and improving oxygen levels, which lowers mortality risk in emergency hospital settings.
- Management of Deep Vein Thrombosis (DVT): Urokinase treats deep vein thrombosis by dissolving clots in deep veins, preventing pulmonary embolism, reducing swelling, and protecting long-term vein health, particularly in severe cases.
- Used During Catheter Blockages: Urokinase clears blood clots in central venous or dialysis catheters, restoring function without surgery, reducing infection risk, and allowing continued treatment in hospitals or home care programs.
- Emergency Use in Acute Ischemic Stroke: Urokinase dissolves clots in acute ischemic stroke within hours of onset, restoring blood flow, limiting brain damage, improving recovery, and providing a cost-effective option in emergency care.
Frequently Asked Questions on Urokinase for Injection
- What is urokinase for injection and how does it work?
Urokinase for injection is a thrombolytic agent that promotes the breakdown of blood clots by converting plasminogen into plasmin. It is administered intravenously in controlled clinical settings to manage thromboembolic conditions effectively. - What are the common indications for urokinase use?
Urokinase is primarily used for the treatment of acute myocardial infarction, pulmonary embolism, deep vein thrombosis, and other thromboembolic disorders. It helps dissolve clots rapidly to restore normal blood flow and reduce the risk of complications. - How should urokinase for injection be stored?
The product should be stored as a sterile, lyophilized preparation under refrigerated conditions, protected from light. Proper storage maintains enzymatic activity and ensures stability until reconstitution and administration in medical facilities. - Are there any precautions or contraindications?
Urokinase should be used with caution in patients with bleeding disorders, recent surgeries, or active hemorrhage. Healthcare providers carefully evaluate risks and monitor patients closely to minimize adverse events during thrombolytic therapy. - Which regions dominate the urokinase market?
North America holds the largest market share of 40.2% due to high thromboembolic disorder prevalence, an aging population, and advanced healthcare infrastructure. Asia Pacific is expected to grow fastest, supported by expanding healthcare access and awareness. - Which product types lead the market?
The market is segmented into powder and solution forms, with the solution type leading at 68.5% share in 2023. Its preference is due to ease of administration and rapid onset of action in clinical thrombolytic treatments. - What are the primary applications driving market growth?
Hospitals dominate the market with a 64.3% share, followed by clinics and other healthcare settings. Growing adoption in hospitals is attributed to higher patient volumes, access to advanced therapies, and established protocols for managing acute thromboembolic events.
Conclusion
In conclusion, urokinase for injection remains a critical thrombolytic therapy addressing a wide range of thromboembolic disorders, including pulmonary embolism, deep vein thrombosis, and acute ischemic stroke. The market is driven by rising disease prevalence, an aging population, and increasing adoption in hospitals, particularly in North America, while Asia Pacific demonstrates the fastest growth due to expanding healthcare infrastructure and awareness.
Emerging trends, including targeted cancer research, safer formulations, tuberculosis applications, and cost-effective therapy, highlight its evolving clinical potential. Continued focus on quality, accessibility, and innovation ensures urokinase’s sustained relevance in global thrombolytic treatment strategies.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



