Table of Contents
Overview
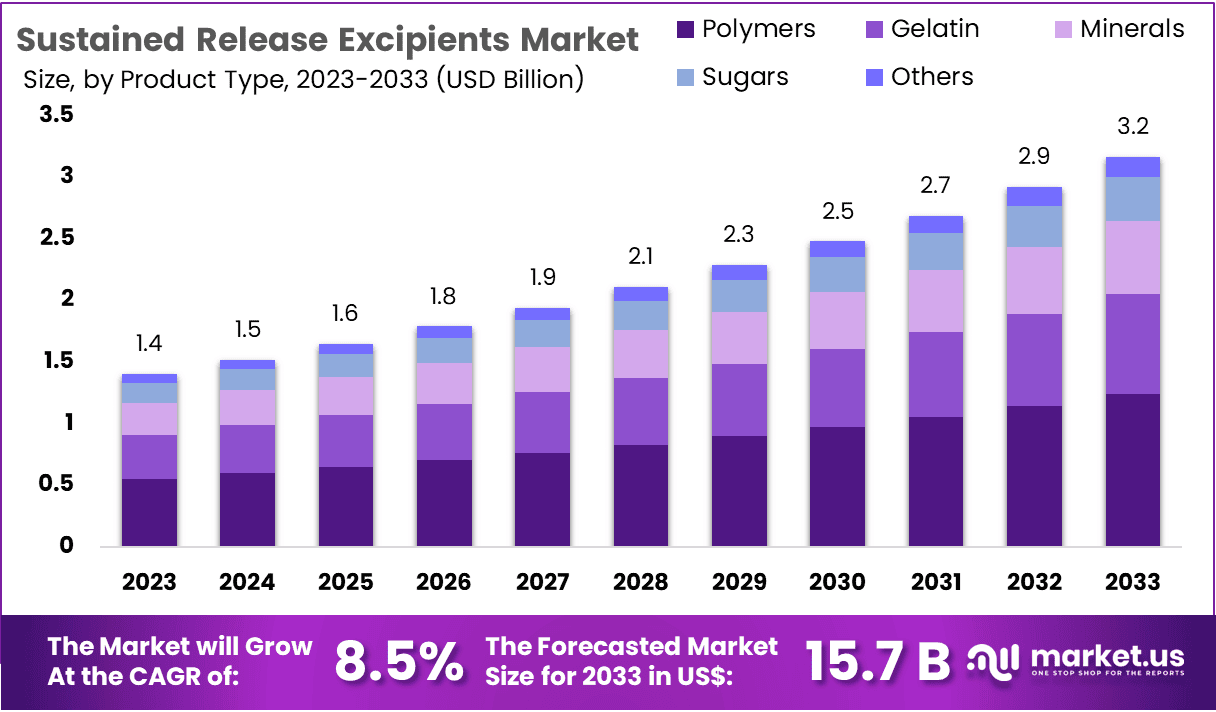
New York, NY – March 06, 2026 – The Global Sustained Release Excipients Market size is expected to be worth around US$ 3.2 Billion by 2033, from US$ 1.4 Billion in 2023, growing at a CAGR of 8.5% during the forecast period from 2024 to 2033. North America dominated the market with a share exceeding 42.4%, achieving a market value of US$ 0.6 billion for the year.
The development of sustained release excipients has become a critical component in modern pharmaceutical formulation. These excipients are specifically designed to control the rate and duration of drug release, enabling consistent therapeutic effects over an extended period. The basic formation of sustained release excipients typically involves the integration of polymers, matrix formers, and release modifiers that regulate the diffusion and dissolution of active pharmaceutical ingredients (APIs).
Commonly used polymers such as hydrophilic cellulose derivatives, acrylic polymers, and biodegradable materials play a key role in establishing controlled drug release profiles. When combined with appropriate binders, fillers, and lubricants, these excipients create a matrix system that gradually releases the drug as the formulation interacts with gastrointestinal fluids. This mechanism helps maintain stable plasma drug concentrations while reducing dosing frequency.
The formulation process generally includes blending the API with selected excipients, followed by granulation and compression into tablets or encapsulation. The structural integrity of the matrix ensures predictable drug diffusion, swelling, or erosion-based release mechanisms depending on the formulation design.
The growing demand for sustained release technologies is driven by the need to improve patient compliance, minimize side effects, and enhance therapeutic efficiency. As pharmaceutical innovation continues to evolve, sustained release excipients are expected to play an increasingly important role in advanced drug delivery systems, supporting the development of safer and more effective medications.
Key Takeaways
- In 2023, the Sustained Release Excipients market generated revenue of US$ 1.4 billion and is projected to reach US$ 3.2 billion by 2033, registering a CAGR of 8.5% during the forecast period.
- Based on product type, the market is segmented into polymers, gelatin, minerals, sugars, and others. Among these, polymers dominated the segment in 2023, accounting for a 39.2% market share.
- By route of administration, the market is categorized into oral, subcutaneous, transdermal, intravenous, and others, with the oral segment holding the largest share of 48.9% in 2023.
- In terms of technology, the market is divided into microencapsulation, targeted delivery, wurster technique, and others. The targeted delivery segment emerged as the leading technology, capturing a 37.4% revenue share in the global market.
- North America dominated the global Sustained Release Excipients market in 2023, accounting for a 42.4% share.
Regional Analysis
North America accounted for the largest share of the Sustained Release Excipients market in 2023, representing 42.4% of total revenue. Market expansion has been supported by continuous advancements in pharmaceutical formulations and rising demand for controlled-release medications. The increasing preference for once-daily dosage forms, which improve patient compliance and treatment convenience, has significantly contributed to market growth.
The rising incidence of chronic conditions such as hypertension, diabetes, and neurological disorders has further accelerated the adoption of sustained release drug formulations. In addition, strong research and development activities focused on improving drug bioavailability and stability, along with supportive regulatory frameworks, have strengthened regional market growth.
The Asia Pacific region is projected to register the fastest CAGR during the forecast period. Growth is being driven by expanding healthcare infrastructure, increasing prevalence of chronic diseases, and improvements in pharmaceutical manufacturing capabilities. Rising disposable income levels, greater awareness of advanced drug delivery technologies, and increasing production of cost-effective generic medicines are also expected to support regional market expansion.
Emerging Trends
- Shift Toward Patient-Centric Drug Formulations: Pharmaceutical companies are increasingly developing patient-centric medicines. Sustained release excipients enable drugs to work longer, reducing dosing frequency. This improves treatment convenience and adherence, particularly for chronic disease patients requiring continuous medication management.
- Increased Use of Natural and Biodegradable Polymers: The use of natural and biodegradable polymers is increasing in pharmaceutical formulations. Materials such as cellulose derivatives offer safer long-term use and improved environmental sustainability, encouraging manufacturers to adopt eco-friendly excipients.
- Advancements in Drug Delivery Technologies: Modern drug delivery technologies such as microspheres, matrix tablets, and multilayer formulations require advanced excipients. Sustained release excipients enable controlled drug release, supporting the development of innovative and highly effective pharmaceutical delivery systems.
- Growing Role in Personalized Medicine: Personalized medicine requires precise drug delivery tailored to individual patient needs. Sustained release excipients help regulate drug release rates, improving treatment effectiveness and safety while supporting customized pharmaceutical formulations.
- Expansion of Oral and Injectable SR Formulations: Applications of sustained release excipients are expanding beyond oral tablets to include long-acting injectable formulations. These systems support extended therapeutic action, reduce administration frequency, and improve treatment management.
- Regulatory Push for Consistency and Safety: Regulatory authorities are emphasizing safety, consistency, and quality in pharmaceutical ingredients. As a result, manufacturers are increasingly adopting well-tested sustained release excipients that meet strict regulatory standards.
Use Cases
- Chronic Disease Treatment: Sustained release excipients are widely used in medications for chronic diseases such as diabetes and hypertension. These excipients maintain stable drug levels in the body and reduce dosing frequency.
- Pain Management Medications: Pain management therapies frequently utilize sustained release excipients to provide prolonged relief. Controlled drug release helps maintain consistent therapeutic levels, reducing the need for repeated dosing.
- Mental Health and Neurological Disorders: Medications for mental health and neurological disorders require stable drug delivery. Sustained release excipients ensure consistent drug levels, improving treatment reliability and minimizing fluctuations that may affect patient outcomes.
- Pediatric and Geriatric Medications: Children and elderly patients often face challenges with frequent medication schedules. Sustained release excipients simplify treatment regimens by enabling longer-acting formulations that reduce the number of doses required.
- Veterinary Medicine: In veterinary healthcare, sustained release excipients support long-acting medications for animals. These formulations reduce dosing frequency, making treatment easier for veterinarians, pet owners, and livestock managers.
- Oncology Therapies: In oncology treatments, sustained release excipients enable gradual release of chemotherapy drugs. This approach helps maintain consistent drug levels, potentially reducing treatment side effects and improving overall therapy management.
Frequently Asked Questions on Sustained Release Excipients
- Why are Sustained Release Excipients important in pharmaceutical formulations?
Sustained release excipients play a critical role in improving drug delivery efficiency by regulating the dissolution and absorption of active pharmaceutical ingredients. Their use enables prolonged therapeutic effects, minimizes side effects, and supports the development of advanced oral and injectable drug delivery systems. - What types of Sustained Release Excipients are commonly used?
Common sustained release excipients include polymers, lipids, and hydrophilic matrix agents such as cellulose derivatives and acrylic polymers. These materials form matrices or coatings that gradually release the drug, ensuring controlled therapeutic activity over a specified treatment duration. - What factors influence the performance of Sustained Release Excipients?
The effectiveness of sustained release excipients depends on polymer composition, drug solubility, formulation design, and environmental conditions such as pH and temperature. These parameters influence drug diffusion, matrix erosion, and overall release kinetics in pharmaceutical dosage forms. - What are the key growth drivers of the Sustained Release Excipients Market?
Market growth is primarily driven by increasing demand for controlled drug delivery systems, rising chronic disease prevalence, and growing pharmaceutical research activities. Additionally, innovation in polymer technology and expanding generic drug manufacturing are contributing to sustained market expansion globally. - Which industries primarily use Sustained Release Excipients?
Sustained release excipients are primarily utilized in the pharmaceutical and biotechnology industries. They are extensively used in oral tablets, capsules, injectable formulations, and transdermal systems to enable prolonged therapeutic effectiveness and improve overall treatment efficiency. - Which regions dominate the Sustained Release Excipients Market?
North America and Europe hold significant market shares due to advanced pharmaceutical manufacturing capabilities and strong research infrastructure. Meanwhile, Asia-Pacific is witnessing accelerated growth, supported by expanding pharmaceutical production and increasing investment in drug development activities. - What is the future outlook for the Sustained Release Excipients Market?
The sustained release excipients market is expected to experience steady growth in the coming years. Increasing focus on patient-centric drug delivery, technological advancements in polymer science, and growing investment in pharmaceutical innovation are anticipated to support long-term market development.
Conclusion
The sustained release excipients market is experiencing steady growth, supported by increasing demand for advanced drug delivery systems and improved pharmaceutical formulations. These excipients play a crucial role in controlling drug release, enhancing therapeutic effectiveness, and improving patient compliance through reduced dosing frequency.
Strong research and development activities, particularly in polymer technology and targeted delivery systems, are further strengthening market expansion. North America currently leads the market, while Asia Pacific is emerging as a high-growth region due to expanding pharmaceutical manufacturing capabilities. With rising chronic disease prevalence and growing adoption of patient-centric treatments, sustained release excipients are expected to remain essential in future drug development.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



