Table of Contents
Overview
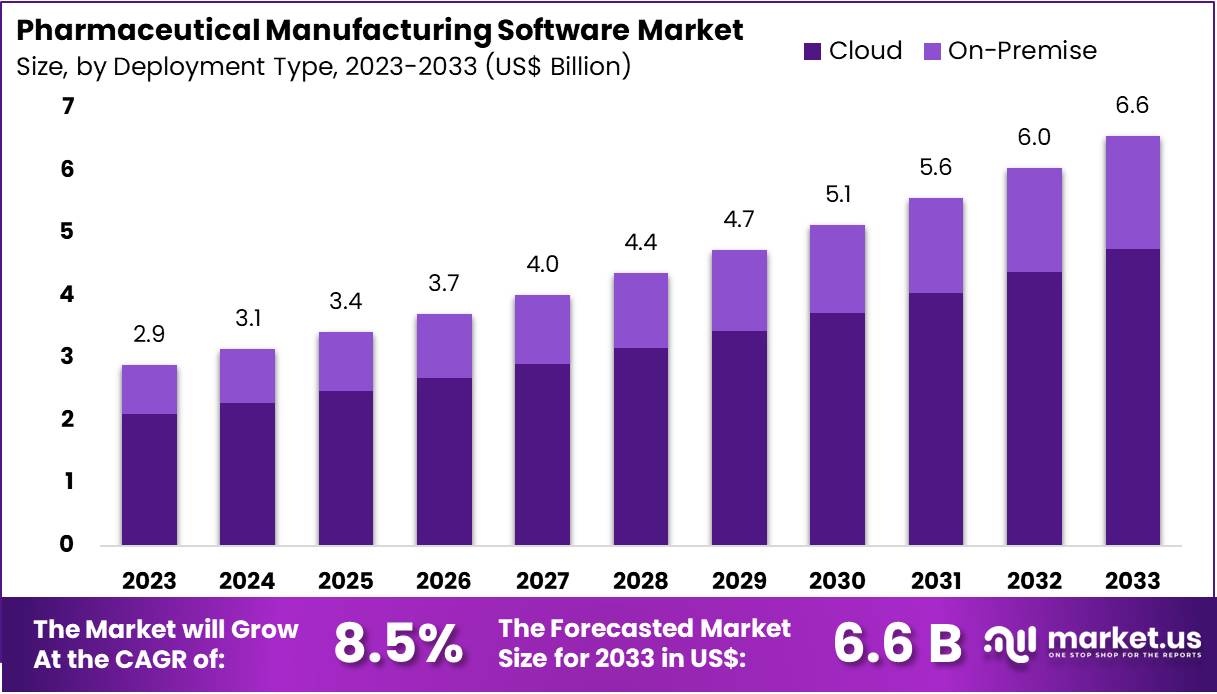
New York, NY – March 06, 2026 – The Global Pharmaceutical Manufacturing Software Market size is expected to be worth around US$ 6.6 Billion by 2033, from US$ 2.9 Billion in 2023, growing at a CAGR of 8.5% during the forecast period from 2024 to 2033. North America held a dominant market position, capturing more than a 40.7% share and holds US$ 1.2 Billion market value for the year.
Pharmaceutical Manufacturing Software plays a critical role in improving efficiency, compliance, and quality control within modern pharmaceutical production facilities. The software is designed to support key manufacturing operations such as batch management, production planning, quality monitoring, inventory control, and regulatory documentation. With increasing regulatory requirements and growing demand for high-quality medicines, pharmaceutical manufacturers are adopting advanced digital solutions to streamline operations and reduce operational risks.
The basic formation of pharmaceutical manufacturing software typically includes several integrated modules. Manufacturing Execution Systems (MES) enable real-time monitoring of production processes and ensure adherence to standardized manufacturing protocols. Enterprise Resource Planning (ERP) components assist in managing supply chain activities, procurement, and financial operations.
Quality Management Systems (QMS) are incorporated to maintain compliance with regulatory standards and to monitor product quality throughout the production cycle. In addition, data analytics tools provide insights that support decision-making and process optimization.
The adoption of pharmaceutical manufacturing software has been steadily increasing as companies focus on automation, digital transformation, and regulatory compliance. These solutions support improved production accuracy, enhanced traceability, and better resource utilization across pharmaceutical manufacturing environments.
Key Takeaways
- In 2023, the Pharmaceutical Manufacturing Software market generated revenue of US$ 2.9 billion and is projected to grow at a CAGR of 8.5%, reaching approximately US$ 6.6 billion by 2033.
- Based on deployment type, the market is categorized into on-premise and cloud solutions. The cloud segment dominated in 2023, accounting for a 72.5% market share.
- In terms of application, the market is segmented into large enterprises and small & medium-sized enterprises (SMEs). Large enterprises held the leading position, representing 67.9% of the total market share in 2023.
- Regarding end users, biopharmaceutical companies emerged as the largest segment, contributing 42.3% of the total revenue share in the Pharmaceutical Manufacturing Software market.
- North America remained the leading regional market, capturing 40.7% of the global market share in 2023.
Regional Analysis
North America Leads the Pharmaceutical Manufacturing Software Market
North America accounted for the largest share of 40.7% in 2023 in the Pharmaceutical Manufacturing Software market. Market leadership has been supported by strong technological advancements and the growing need for efficient, automated drug manufacturing processes. Pharmaceutical companies across the region are increasingly adopting digital platforms to improve production efficiency, maintain regulatory compliance, and optimize operational costs.
The integration of technologies such as artificial intelligence, automation, and data analytics has strengthened the adoption of advanced manufacturing software solutions. Strategic collaborations are also contributing to innovation; for instance, a partnership between Amazon Web Services and Pfizer was established to develop cloud-based solutions for drug development and manufacturing.
Asia Pacific Expected to Record the Fastest Growth
The Asia Pacific region is projected to register the highest growth during the forecast period, driven by expanding pharmaceutical production and increasing digital investments. Countries such as China and India are strengthening manufacturing capabilities through advanced technologies. Collaborative initiatives, including partnerships between Triastek, Inc. and Siemens Ltd., are accelerating the adoption of digital manufacturing solutions across the region.
Use Cases
- Production Planning and Scheduling: Pharmaceutical manufacturing software supports production planning and scheduling by organizing manufacturing tasks efficiently. It helps allocate machines, workforce, and materials properly, reducing delays, improving workflow coordination, and ensuring timely completion of pharmaceutical production cycles.
- Quality Control and Batch Management: Manufacturing software enables pharmaceutical companies to track each production batch with complete traceability. It records quality testing results, identifies deviations quickly, and ensures that every batch meets required safety and quality standards.
- Regulatory Reporting and Documentation: Pharmaceutical manufacturing software automates documentation and reporting processes required for regulatory compliance. It stores operational data, generates reports, and maintains accurate records, allowing companies to respond efficiently to inspections, audits, and regulatory requirements.
- Inventory and Material Tracking: The software helps manufacturers track raw materials, components, and finished products in real time. Inventory alerts, lot tracking, and expiration monitoring improve material management, reduce waste, and ensure continuous availability of essential production resources.
- Equipment Maintenance and Monitoring: Pharmaceutical manufacturing software supports preventive maintenance by tracking equipment performance and scheduling servicing activities. Real-time monitoring helps detect early signs of malfunction, reducing downtime, extending equipment lifespan, and ensuring uninterrupted production operations.
- Electronic Batch Records (EBR): Electronic Batch Records allow pharmaceutical companies to digitally document each production stage. The system records material usage, processing steps, and operator actions, improving traceability, minimizing documentation errors, and supporting faster regulatory audits and inspections.
- Collaboration Across Departments: Manufacturing software connects departments such as production, quality assurance, and regulatory teams within a unified platform. Shared data access improves communication, accelerates decision-making, reduces operational delays, and enhances coordination across pharmaceutical manufacturing processes.
Frequently Asked Questions on Pharmaceutical Manufacturing Software
- Why is Pharmaceutical Manufacturing Software important for the pharmaceutical industry?
Pharmaceutical Manufacturing Software improves operational efficiency by automating manufacturing workflows and ensuring compliance with strict regulatory standards. It enhances traceability, minimizes production errors, and supports consistent product quality, which is essential for maintaining safety and reliability in pharmaceutical manufacturing environments. - What are the key features of Pharmaceutical Manufacturing Software?
Key features typically include batch management, quality assurance tracking, electronic batch records, real-time production monitoring, regulatory compliance tools, and supply chain integration. These functionalities enable manufacturers to streamline production processes while maintaining strict adherence to industry regulations and quality standards. - What factors are driving growth in the Pharmaceutical Manufacturing Software Market?
Growth in the Pharmaceutical Manufacturing Software Market is driven by increasing regulatory requirements, rising demand for pharmaceutical products, and growing adoption of digital manufacturing technologies. Pharmaceutical companies are increasingly implementing advanced software systems to improve operational efficiency, compliance management, and production transparency. - Who are the primary users of Pharmaceutical Manufacturing Software?
Primary users include pharmaceutical manufacturers, biotechnology companies, contract manufacturing organizations, and research laboratories. These organizations rely on specialized software solutions to manage complex manufacturing workflows, maintain regulatory compliance, and improve overall production efficiency in drug manufacturing facilities. - How does Pharmaceutical Manufacturing Software help ensure regulatory compliance?
Pharmaceutical Manufacturing Software supports compliance by maintaining detailed production records, automating documentation, and ensuring traceability across manufacturing processes. Built-in validation tools and audit trails assist companies in meeting regulatory standards established by global health authorities and pharmaceutical regulatory agencies. - What deployment models are commonly used in Pharmaceutical Manufacturing Software?
Common deployment models include on-premise and cloud-based solutions. On-premise systems offer enhanced data control and customization, while cloud-based platforms provide scalability, remote accessibility, and lower infrastructure costs, making them increasingly attractive for modern pharmaceutical manufacturing environments. - Which regions are leading in the Pharmaceutical Manufacturing Software Market?
North America and Europe are considered leading regions due to advanced pharmaceutical industries, strong regulatory frameworks, and early adoption of digital manufacturing technologies. Meanwhile, Asia-Pacific is experiencing rapid growth driven by expanding pharmaceutical production and increasing technology investments.
Conclusion
The Pharmaceutical Manufacturing Software sector continues to gain strategic importance as pharmaceutical companies accelerate digital transformation across production environments. Growing regulatory requirements, increasing demand for high-quality medicines, and the need for operational efficiency are driving widespread software adoption.
Advanced platforms integrating manufacturing execution, quality management, data analytics, and supply chain visibility are improving traceability and production accuracy. Market expansion is supported by strong adoption in North America and rapid technological investments in Asia Pacific. With continued advancements in automation, artificial intelligence, and cloud deployment, the Pharmaceutical Manufacturing Software Market is expected to experience steady growth and enhanced industry integration.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



