Table of Contents
Overview
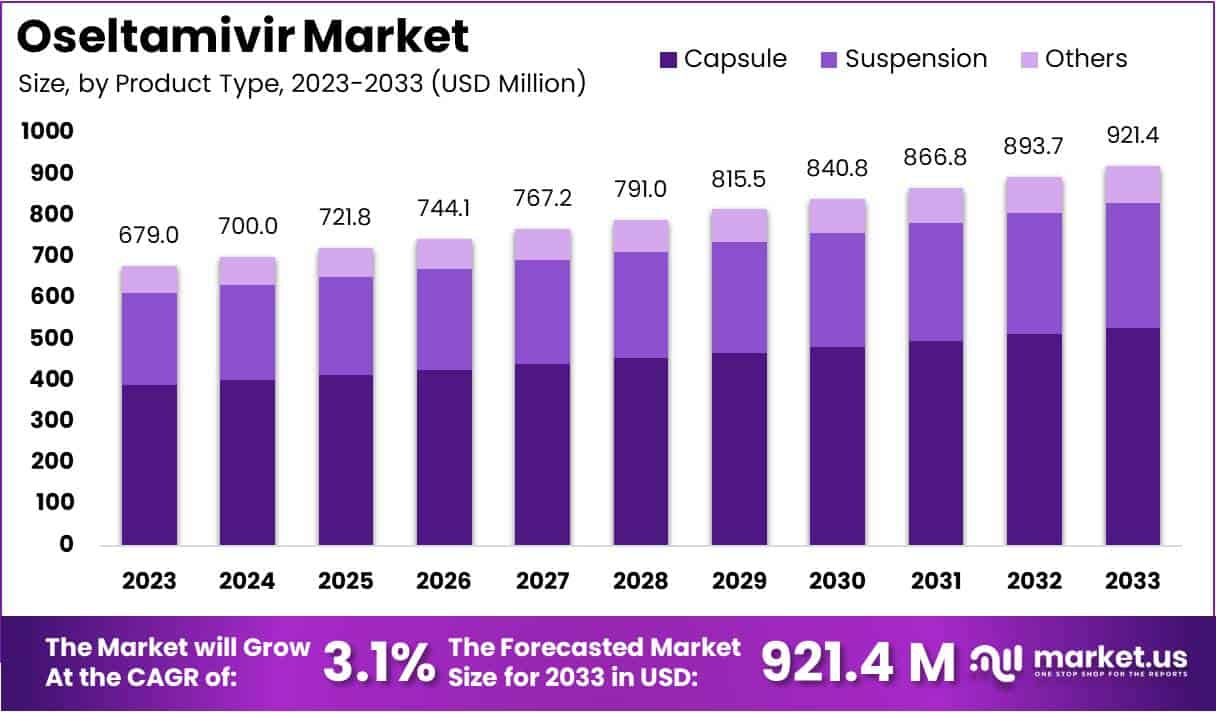
New York, NY – March 04, 2026 – The Global Oseltamivir Market size is expected to be worth around US$ 921.4 Million by 2033 from US$ 679 Million in 2023, growing at a CAGR of 3.1% during the forecast period 2024 to 2033.
Oseltamivir is a widely utilized antiviral agent indicated for the treatment and prevention of influenza A and influenza B infections. Commercially recognized under the brand name Tamiflu, the compound has been extensively adopted in seasonal and pandemic influenza management protocols across global healthcare systems.
Chemically, oseltamivir is administered as oseltamivir phosphate, a prodrug that undergoes hepatic conversion to its active metabolite, oseltamivir carboxylate. The active form selectively inhibits neuraminidase enzymes present on the surface of influenza viruses. By blocking neuraminidase activity, viral replication and release from infected host cells are significantly reduced. This mechanism limits viral spread within the respiratory tract and supports faster symptom resolution when treatment is initiated within 48 hours of onset.
The basic molecular framework of oseltamivir consists of a cyclohexene core structure substituted with functional groups that enhance oral bioavailability and systemic absorption. The phosphate salt formulation improves stability, solubility, and ease of large-scale manufacturing. Standard oral dosage forms include capsules and powder for oral suspension, supporting pediatric and adult administration.
Global demand for oseltamivir has been driven by recurrent influenza outbreaks, pandemic preparedness strategies, and stockpiling initiatives by public health authorities. Ongoing research and manufacturing advancements continue to strengthen supply chain resilience and ensure therapeutic accessibility in both developed and emerging markets.
Key Takeaways
- In 2023, the Oseltamivir market generated revenue of US$ 679 million and is expanding at a compound annual growth rate (CAGR) of 3.1%.
- The market valuation is projected to reach approximately US$ 921.4 million by 2033, reflecting steady long-term demand.
- By product type, the market is segmented into suspension, capsules, and other formulations, with capsules accounting for the largest share of 57.4% in 2023.
- Based on application, the market is categorized into influenza A, influenza B, and other indications, with influenza A representing 49.6% of the total market share in 2023.
- Regionally, North America led the global Oseltamivir market, capturing a dominant share of 39.4% in 2023.
Regional Analysis
North America accounted for the largest share of the Oseltamivir market, representing 39.4% of global revenue in 2023. Market dominance has been supported by elevated influenza activity and structured governmental preparedness initiatives.
In December 2022, the U.S. Department of Health and Human Services, through the Administration for Strategic Preparedness and Response, released additional supplies of Tamiflu from the national stockpile to address rising antiviral demand. This measure ensured sufficient product availability across states during peak flu season. Strong public health campaigns promoting early antiviral treatment and continuous strategic stockpiling further sustained regional demand.
Asia Pacific is projected to register the fastest CAGR over the forecast period. Growth is being driven by rising influenza cases, expanding healthcare infrastructure, and strengthened distribution agreements. In 2023, Zhongchao Inc. announced a Mainland China distribution partnership involving Natco Pharma Limited, enhancing antiviral accessibility. Improved diagnostics and government-led infectious disease management programs are expected to support continued market expansion.
Emerging Trends
- Rising Focus on Pediatric Formulations: Pharmaceutical manufacturers are increasingly developing pediatric-friendly oseltamivir formulations, including flavored suspensions and low-dose capsules. These innovations improve treatment adherence among children, enhance ease of administration at home, and support more effective influenza management in younger populations.
- Increased Government Stockpiling: Governments are strengthening antiviral stockpiling strategies to address seasonal influenza outbreaks and potential pandemics. Strategic reserves of oseltamivir enable rapid distribution during health emergencies, ensuring timely treatment access and reinforcing national preparedness frameworks against infectious disease threats.
- Development of Extended-Release Versions: Pharmaceutical innovation is advancing toward extended-release oseltamivir formulations designed to reduce dosing frequency. Once-daily administration improves patient compliance, enhances therapeutic outcomes, and may minimize side effects, thereby increasing overall treatment efficiency during influenza seasons.
- Wider Global Accessibility Through Generics: The expansion of generic oseltamivir production has improved affordability and accessibility, particularly in low- and middle-income countries. Competitive pricing and diversified manufacturing have strengthened supply stability, supporting broader influenza treatment coverage across underserved healthcare systems.
Use Cases
- Seasonal Influenza Treatment in Adults and Children: Oseltamivir is widely prescribed for early-stage influenza treatment in adults and children. Administration within 48 hours reduces illness duration, lowers complication risks, and supports faster recovery, particularly during peak seasonal outbreaks.
- Preventive Use in Flu Outbreaks: Oseltamivir is utilized for post-exposure prophylaxis among individuals at risk of influenza infection. Preventive administration significantly reduces transmission rates in households and institutional settings, protecting vulnerable populations during localized outbreaks.
- Support During Flu Pandemics: During influenza pandemics, including the H1N1 influenza outbreak, oseltamivir played a critical therapeutic and preventive role. Its inclusion in national preparedness plans strengthened outbreak response, reduced transmission risks, and supported frontline healthcare systems globally.
- Treatment for High-Risk Patients: High-risk individuals with chronic conditions such as asthma, diabetes, or cardiovascular disease benefit significantly from early oseltamivir treatment. Prompt administration reduces hospitalization risk, mitigates severe complications, and improves clinical outcomes in vulnerable patient groups.
Frequently Asked Questions on Oseltamivir
- How does Oseltamivir work?
Oseltamivir functions by inhibiting the neuraminidase enzyme present on the influenza virus surface. This mechanism prevents the release of newly formed viral particles from infected cells, thereby reducing viral spread within the respiratory tract and supporting faster recovery. - What are the primary indications for Oseltamivir?
Oseltamivir is primarily indicated for the treatment of acute, uncomplicated influenza in adults and pediatric patients. It is also recommended for post-exposure prophylaxis in high-risk populations, including elderly individuals and patients with chronic health conditions. - What are the common side effects of Oseltamivir?
Common adverse effects associated with Oseltamivir include nausea, vomiting, headache, and mild gastrointestinal discomfort. These effects are generally transient and resolve without intervention. Rare neuropsychiatric events have been reported, particularly among pediatric populations. - What factors are driving the growth of the Oseltamivir market?
Growth of the Oseltamivir market is driven by increasing seasonal influenza prevalence, rising awareness regarding antiviral therapies, and government stockpiling initiatives. Expansion of healthcare infrastructure in emerging economies has also contributed to higher demand and broader product accessibility. - Which regions dominate the Oseltamivir market?
North America and Europe hold significant shares in the Oseltamivir market due to strong healthcare systems and established vaccination programs. However, Asia-Pacific is projected to witness accelerated growth, supported by large populations and improving medical access. - Who are the key players in the Oseltamivir market?
The Oseltamivir market is characterized by the presence of multinational pharmaceutical companies and regional generic manufacturers. Market competition is influenced by patent expirations, production capacity, distribution networks, and strategic collaborations within the antiviral drug segment. - What is the future outlook of the Oseltamivir market?
The Oseltamivir market is expected to demonstrate steady growth, supported by recurring influenza outbreaks and public health preparedness initiatives. Continuous research on antiviral resistance and formulation improvements is anticipated to sustain long-term commercial viability.
Conclusion
Oseltamivir, commercially marketed as Tamiflu, remains a critical antiviral therapy for managing influenza A and B across global healthcare systems. Market performance reflects stable growth, supported by recurring seasonal outbreaks, structured stockpiling programs, and expanding generic penetration.
North America maintains revenue leadership, while Asia Pacific is projected to record the fastest expansion due to infrastructure development and distribution partnerships. Product innovation, including pediatric and extended-release formulations, is enhancing patient compliance. With revenues expected to reach US$ 921.4 million by 2033, sustained demand is anticipated under evolving public health preparedness strategies.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



