Table of Contents
Introduction
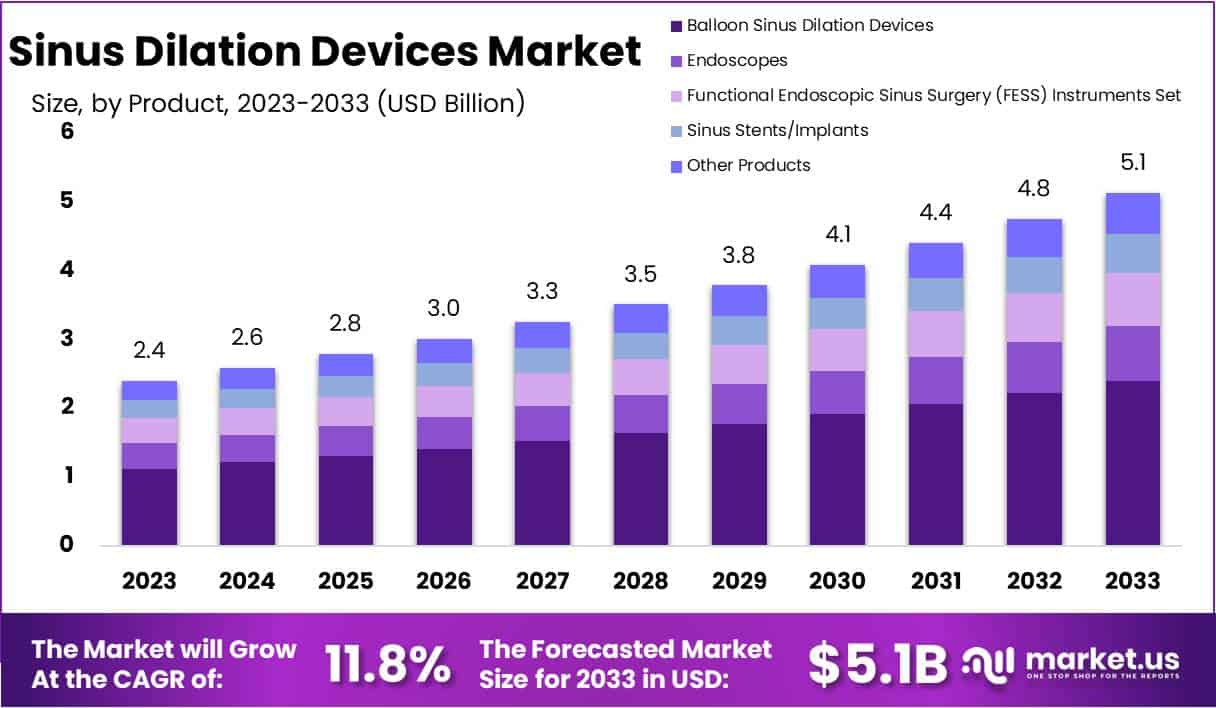
The Global Sinus Dilation Devices Market is projected to reach USD 5.1 billion by 2033, up from USD 2.4 billion in 2023, growing at a CAGR of 11.8% during the forecast period (2024-2033). This growth is driven by increasing cases of chronic rhinosinusitis (CRS), a rising preference for minimally invasive procedures, and advancements in sinus treatment technology. The market is segmented by product type, procedure, application, end-use, and region, with hospitals and ENT clinics playing a major role in adoption.
CRS affects 10-15% of the global population, increasing the demand for effective treatments. Pollution, allergies, and respiratory infections are major contributors. Balloon sinuplasty, a minimally invasive alternative to traditional sinus surgery, is gaining traction due to its effectiveness in improving patient outcomes with shorter recovery times.
Sinus dilation procedures are preferred over conventional surgeries due to faster recovery, reduced complications, and lower costs. These procedures are often performed in outpatient settings, eliminating hospital stays. Governments and healthcare providers are promoting cost-effective treatments, further boosting adoption.
The market is witnessing significant technological advancements, including navigation-assisted procedures, enhanced balloon catheter designs, and single-use devices. The introduction of robotic-assisted sinus surgeries is expected to further drive demand in the coming years.
Developing nations are investing in modern ENT clinics and hospitals, improving access to sinus treatments. Rural healthcare initiatives and increased government spending on medical devices are further propelling market growth. Developed regions are also witnessing rapid adoption due to higher healthcare budgets.
Regulatory bodies like the U.S. FDA and the EMA have approved several sinus dilation devices, ensuring safety and efficiency. Streamlined approval processes encourage new product launches and investments from medical device companies. Additionally, increased awareness campaigns by the WHO, CDC, and national health agencies are leading to early diagnosis and higher patient visits to ENT specialists.
The sinus dilation devices market is set for strong growth, driven by rising CRS cases, demand for minimally invasive treatments, and continuous innovation in medical technology. Increasing healthcare infrastructure and favorable regulatory policies are further boosting adoption. As healthcare systems evolve, the demand for effective, cost-efficient sinus treatments will continue to expand, ensuring steady market progression.
Key Takeaways
- Market Growth: The market is projected to reach USD 5.1 billion by 2033, expanding at a CAGR of 11.8% from 2024 to 2033.
- Product Dominance: Balloon Sinus Dilation Devices held a 46.8% market share in 2023, reflecting their widespread adoption due to effectiveness and minimal invasiveness.
- Procedure Preference: Standalone procedures accounted for 67.4% of the market in 2023, highlighting a preference for simplified, efficient sinus treatment options.
- Target Demographics: The adult segment led with 67.9% share in 2023, indicating a higher prevalence of sinus-related conditions among adults compared to younger demographics.
- End-Use Leadership: Hospitals dominated the market with a 53.1% share in 2023, reflecting their advanced facilities and expertise in sinus dilation procedures.
- Market Drivers: A global rise in chronic sinusitis cases, coupled with technological advancements and increased awareness, is fueling demand for sinus dilation devices.
- Market Restraints: High procedural costs and strict regulatory requirements limit market accessibility, posing challenges to widespread adoption and growth.
- Growth Opportunities: The expanding geriatric population and improving healthcare infrastructure in emerging markets present lucrative opportunities for market expansion.
- Key Trends: Balloon sinuplasty adoption is rising, and industry collaborations are driving innovation in product development and treatment approaches.
- Regional Dominance: North America held 40.1% market share in 2023, driven by high sinusitis prevalence and advanced healthcare infrastructure supporting sinus treatments.
Emerging Trends
- Preference for Minimally Invasive Procedures: Patients now prefer treatments that cause less discomfort and speed up recovery. Balloon sinuplasty is a key example. It uses small, flexible tools inserted through the nostrils to open blocked sinus passages. Unlike traditional surgery, it does not require tissue or bone removal. This technique preserves the natural sinus structure and reduces complications. Many patients can also undergo this procedure in a doctor’s office with only local anesthesia. The convenience and effectiveness of balloon sinuplasty have increased its adoption among both patients and healthcare providers.
- Technological Advancements in Navigation Systems: New microsensor technology has improved the precision of sinus dilation procedures. Flexible guidewires equipped with sensors help doctors track their movements in real time. This makes navigation through the complex sinus anatomy safer and more effective. With better accuracy, there is a lower risk of damaging surrounding tissues. These technological improvements have made balloon sinus ostial dilation more reliable. As a result, patient outcomes have improved, and the procedure has become a preferred option for sinus relief.
- Expansion of FDA Approvals: The U.S. Food and Drug Administration (FDA) has expanded approvals for balloon sinuplasty devices. Different devices are now available for various age groups and sinus conditions. For example, one device treats frontal and sphenoid sinus disease in adults but is also approved for maxillary sinus issues in children under 17. Another is approved for maxillary sinuses in children as young as two and for frontal and sphenoid sinuses in patients over 12. These approvals have increased access to sinus dilation procedures, making them available to a wider range of patients.
- Integration with Endoscopic Techniques: Balloon sinuplasty is now frequently combined with endoscopic methods. This integration improves visibility during procedures. Surgeons can use micro-cutting instruments to remove abnormal or obstructive tissue. This enhances sinus drainage and helps prevent future infections. By combining these techniques, doctors achieve better long-term outcomes. Patients also benefit from reduced symptoms and quicker recovery times. The combination of balloon sinuplasty and endoscopic methods is becoming a standard approach in sinus care.
Use Cases
- Treatment of Chronic or Recurrent Sinusitis: Sinus dilation devices help treat chronic or recurring sinus infections that do not improve with medication. These devices work by widening the sinus openings, restoring normal drainage, and reducing blockages. This helps relieve symptoms like nasal congestion, facial pain, and sinus pressure. The procedure is minimally invasive, making it a safer alternative to traditional surgery. Many patients experience long-term relief after treatment. Compared to other surgical options, recovery is faster, with minimal discomfort. This approach is suitable for individuals looking for a non-surgical solution to persistent sinus problems. It can improve the quality of life by reducing the frequency of infections and dependence on medication.
- Pediatric Applications: Certain balloon sinuplasty devices are safe for children with chronic sinusitis. These devices provide a minimally invasive treatment option, reducing the need for more aggressive surgical procedures. Some devices are approved for use in children as young as two years old, particularly for maxillary sinus issues. This expands treatment options for younger patients who struggle with persistent sinus infections. The procedure is quick, with minimal recovery time, making it ideal for pediatric use. It reduces the need for long-term medication, improving the child’s overall health. Parents can consider this option when traditional treatments fail to provide relief.
- Office-Based Procedures: Balloon sinuplasty is a minimally invasive procedure that can be done in a doctor’s office. It only requires local anesthesia, avoiding the risks of general anesthesia. This makes it a convenient option for patients who prefer outpatient care. The procedure is quick, typically lasting less than an hour. Recovery is also faster compared to traditional sinus surgery. Patients can return to normal activities within a day or two. Office-based procedures reduce hospital visits and associated costs, making sinus treatment more accessible. This option is ideal for individuals seeking effective relief without hospital stays.
- Combination with Traditional Endoscopic Sinus Surgery: Sinus dilation devices can be used alongside endoscopic sinus surgery for better results. In some cases, additional intervention is needed to remove obstructive tissue. Combining both methods ensures improved sinus drainage and long-term relief. Balloon sinuplasty can help widen the sinus passages, making the surgical process smoother. This approach enhances the overall success of the treatment. It is often recommended for patients with severe sinus conditions who require a more comprehensive solution. By integrating sinus dilation devices with traditional surgery, doctors can achieve better patient outcomes with minimal complications.
Conclusion
The sinus dilation devices market is experiencing steady growth due to the increasing prevalence of chronic sinusitis and the rising demand for minimally invasive treatments. Advances in technology, such as navigation-assisted procedures and improved balloon catheter designs, are further driving market expansion. Hospitals and ENT clinics continue to be key adopters, benefiting from enhanced patient outcomes and reduced recovery times. Regulatory approvals and government initiatives are improving access to these treatments worldwide. As healthcare infrastructure develops, particularly in emerging markets, the demand for effective and affordable sinus treatments will continue to rise. With ongoing innovations and increasing awareness, the sinus dilation devices market is set for sustained expansion in the coming years.
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



