Table of Contents
Overview
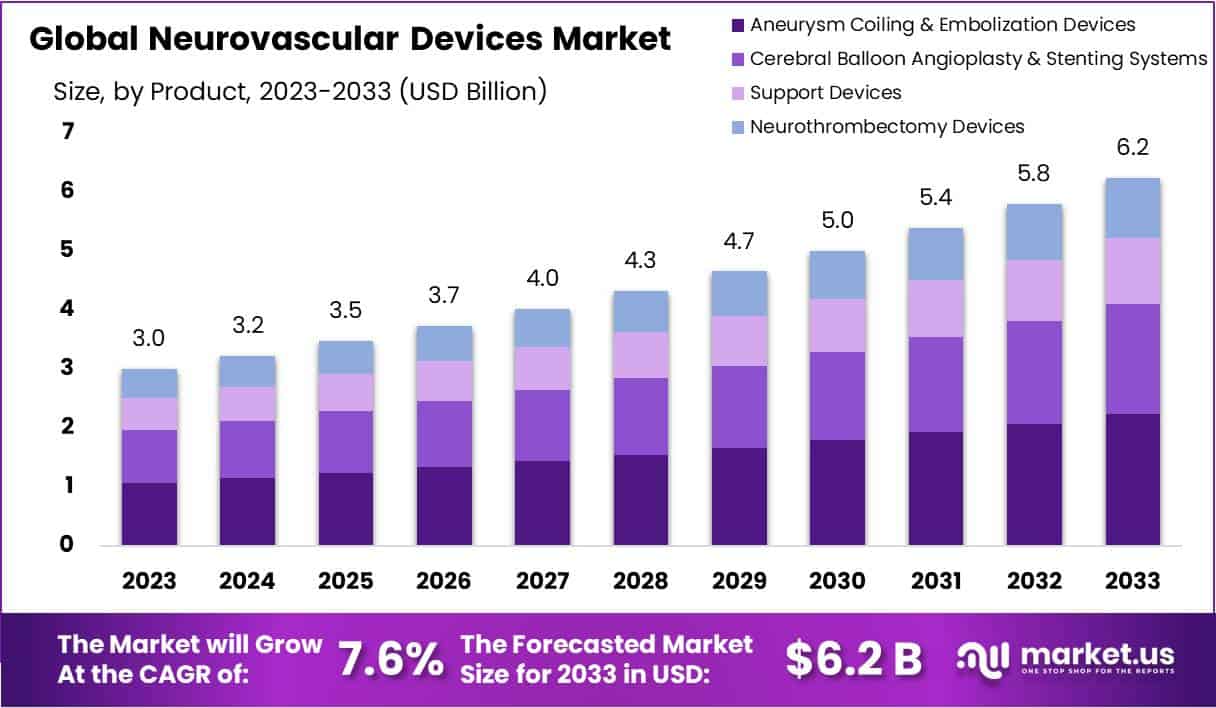
New York, NY – August 29, 2025: The global neurovascular devices market was valued at USD 3 Billion in 2023 and is projected to reach around USD 6.2 Billion by 2033, growing at a CAGR of 7.6% during the forecast period. Growth is driven by the rising prevalence of cerebrovascular diseases, including stroke, aneurysms, and arteriovenous malformations. Stroke remains the second leading cause of death worldwide, creating significant demand for devices such as stents, embolic coils, thrombectomy devices, and flow diverters. The aging global population also contributes to higher risks, boosting the need for advanced treatment solutions.
Technological innovation is another key factor propelling the sector forward. Advancements in clot retrieval systems, embolization devices, and neuro-stents are enhancing treatment outcomes and patient recovery. The integration of robotics, imaging, and AI-assisted navigation has improved precision in neurointerventions. These developments support safer and more efficient procedures, encouraging wider adoption across hospitals and specialized neurology centers. As a result, the demand for modern, minimally invasive neurovascular devices continues to accelerate worldwide, ensuring consistent market expansion in the coming years.
The increasing preference for minimally invasive treatments is further shaping market demand. Endovascular and catheter-based interventions are preferred over open surgeries due to shorter hospital stays, reduced complications, and faster recovery. Growing awareness through public health campaigns and improvements in neuroimaging technologies are enabling early detection of stroke and aneurysms. This, in turn, increases the uptake of neurovascular devices for timely interventions. The trend of early diagnosis and treatment adoption is expanding rapidly across both developed regions and emerging economies, strengthening market penetration globally.
Healthcare infrastructure development and strategic collaborations are also fueling industry growth. Governments and private sector investments in neurology departments and stroke centers are improving access to treatment. Additionally, medical device companies are partnering with research institutions to advance next-generation technologies and accelerate clinical trials. Medical tourism in Asia-Pacific and Latin America is also boosting adoption due to affordable and high-quality care. As accessibility improves worldwide, the neurovascular devices market is set to expand steadily, driven by innovation, demand, and healthcare investments.
Key Takeaways
- The neurovascular devices market is projected to expand at a CAGR of 7.6%, reaching nearly USD 6.2 billion globally by 2033.
- Market growth is largely driven by the increasing prevalence of neurological disorders, continuous technological innovations, and the growing preference for minimally invasive medical treatments.
- Embolization devices and aneurysm-coiling devices currently lead the technology segment, together accounting for approximately 35.7% of the global neurovascular devices market.
- Among disease pathologies, ischemic strokes dominate, emphasizing the critical need for advanced devices targeting brain vessel blockages to improve patient treatment outcomes.
- Stroke treatment remains the most significant application area, holding 58.3% market share in 2023, fueled by rising stroke incidence and widespread hypertension prevalence.
- Neurovascular devices sized at 0.021″ were most preferred in 2023, reflecting healthcare professionals’ inclination toward specific device sizes for effective procedures.
- Hospitals and surgical centers continue to dominate end-user adoption, representing about 70.5% of the market share due to rising patient inflow and specialized care.
- Regionally, North America led the global neurovascular devices market with a 29.2% share in 2023, supported by high diagnosis rates and healthcare spending.
Regional Analysis
In 2023, North America is set to dominate the neurovascular devices market, holding a 29.2% share and a market value of USD 0.87 Billion. The growth is driven by the rising prevalence of neurological disorders and higher diagnosis and treatment rates. Increased healthcare spending further supports neurological care services. Advanced treatment solutions, including innovative neurovascular devices, are likely to gain wider acceptance. Favorable reimbursement policies also encourage adoption, making the region a key hub for market expansion in the coming years.
Europe is expected to secure a significant share of the neurovascular devices market. Market growth is supported by rising awareness of proper neurovascular condition management. Multiple product launches targeted at treating these conditions have strengthened the regional market. Major players are actively expanding their geographic footprint across Europe. These efforts, combined with increasing healthcare investments and patient access to advanced therapies, create favorable conditions for steady growth in the neurovascular devices sector.
The European market is further driven by strategic initiatives from key manufacturers. Companies are focusing on innovation, regulatory approvals, and collaborations to enhance market penetration. Rising demand for minimally invasive and technologically advanced neurovascular treatments also contributes to market development. Patient education and awareness programs encourage early diagnosis and intervention. Overall, Europe’s neurovascular devices market is expected to grow steadily, supported by favorable policies, rising healthcare expenditure, and ongoing technological advancements, positioning the region as a prominent contributor to global market growth.
Segmentation Analysis
Technology Analysis
The Neurovascular Devices market is segmented by technology into aneurysm coiling & embolization devices, cerebral balloon angioplasty and stenting systems, support devices, and neurothrombectomy devices. Among these, embolization and aneurysm-coiling devices dominate, capturing 35.7% market share in 2023. Coil embolization is a minimally invasive procedure for treating aneurysms. A steerable catheter guides the coil from the groin to the brain to reduce blood flow risks. Flow liquid embolic and diversion devices further enhance treatment options, driving segment growth.
Disease Pathology Analysis
Based on disease pathology, the market is divided into ischemic strokes, cerebral aneurysms, carotid artery stenosis, arteriovenous malformations, and fistulas. Ischemic strokes lead the market with 26.8% share. These strokes occur when blood flow to the brain is blocked. Neurovascular devices like stent retrievers and thrombectomy devices restore blood flow. Cerebral aneurysms are treated with coil embolization devices to prevent rupture. Increasing stroke prevalence and aneurysm cases are expected to drive demand for specialized neurovascular treatments in the coming years.
Application Analysis
The Neurovascular Devices market is segmented by application into stroke and cerebral aneurysm treatments. The stroke segment dominated with 58.3% revenue share in 2023. Rising prevalence of hypertension, strokes, and other neurological disorders is fueling growth. In the U.S., strokes cause approximately 140,000 deaths annually and are the second-leading global cause of death. Technological advancements in devices further support segment expansion. Cerebral aneurysm applications are expected to grow due to new product introductions and clinical studies evaluating devices like the Nautilus intravascular system.
Size Analysis
Market segmentation by size shows that 0.021″ devices held the largest share in 2023, around 29.1%. These devices offer benefits such as compatibility with advanced procedures and new product approvals. Recent innovations include Bendit21 microcatheter and Evasc Neurovascular’s eCLIPs Bifurcation Flow Diverter. Devices in the “others” category, including sizes 0.015″, 0.060″, and 0.091″, are expected to grow at the fastest rate. Product development, regulatory approvals, and clinical adoption are key factors driving demand for diverse device sizes across global markets.
End User Analysis
Hospitals dominated the end-user segment with 70.5% share in 2023. Growth is driven by rising patient numbers with neurovascular conditions like strokes, aneurysms, and AVMs. Each year, 13.7 million people experience their first stroke, with 5.4 million deaths. Increasing hospital admissions for treatments, therapies, and surgeries boost device demand. The “other” segment, including ambulatory surgical centers and emergency care facilities, is projected to grow fastest. Expanding healthcare infrastructure, adoption of advanced products, and favorable reimbursement policies contribute to rising end-user demand globally.
Key Players Analysis
Competitors in the neurovascular devices market are heavily focusing on launching and advancing innovative medical devices. In April 2023, MicroVention, Inc., a wholly-owned subsidiary of Terumo Corporation, announced the completion of its initial enrolment into STRAIT. This EU-based multi-center prospective observational study aims to evaluate the safety and performance of the BOBBY Balloon Guide Catheter. The study’s results are expected to provide critical clinical evidence supporting its use in endovascular treatment of acute ischemic stroke, a high-growth therapeutic area.
The BOBBY Balloon Guide Catheter has gained approval in both North America and Europe. It was specifically designed to improve compatibility with the SOFIA 6Fr Aspiration Catheter. Additionally, the device simplifies balloon preparation for clinicians, reducing procedural complexity. These functional advancements enhance navigation through tortuous vessels, making the procedure safer and more efficient. As hospitals adopt innovative neurovascular solutions, the market is expected to witness steady growth over the forecast period, driven by technological improvements.
Market growth is further fueled by ongoing clinical validation and adoption of advanced devices. With STRAIT and similar studies, medical professionals gain confidence in safety and efficacy. This, in turn, accelerates the integration of novel devices in clinical practice. Moreover, improved procedural efficiency and reduced operation time are key factors influencing hospital purchasing decisions. As manufacturers continue to innovate, the neurovascular devices segment is projected to expand. The combination of regulatory approvals, technological compatibility, and positive clinical outcomes supports long-term market growth.
Market Key Players
- Medtronic plc
- Johnson and Johnson Services Inc.
- Penumbra Inc
- Microport Scientific Corporation
- Stryker
- Microvention Inc.
- Codman Neuro
- Abbott
- Terumo Corporation
- Medilit Co. Ltd.
- Evasc
- Rapid Medical
- Other Market Players
Challenges
- High Costs and Limited Accessibility: Neurovascular devices are highly advanced, which makes them expensive. High costs create a significant barrier for hospitals and clinics, especially in low-income countries. Patients in developing regions often cannot afford these treatments, limiting their access to life-saving procedures. Even in developed countries, insurance coverage may not fully offset the cost. This financial challenge slows the adoption of new devices. Manufacturers face difficulty expanding their market because the price can restrict patient reach. As a result, many promising technologies remain underutilized. Improving affordability and accessibility is critical to ensure more patients benefit from these advanced treatments.
- Regulatory Hurdles: Neurovascular devices must meet strict regulatory requirements before entering the market. Companies need to conduct extensive clinical trials to demonstrate safety and effectiveness. The process also requires detailed documentation and compliance with local and international regulations. These procedures are often lengthy and costly, which can delay product launches. Smaller companies may struggle with the financial and logistical demands of approvals. Even minor errors in submissions can result in further delays. Regulatory hurdles ensure patient safety but can slow innovation. For manufacturers, navigating these complex processes is a major challenge that affects timelines and overall market growth.
- Risk of Complications: Using neurovascular devices carries inherent medical risks. Procedures can lead to complications such as bleeding, clot formation, or unexpected reactions. Even experienced surgeons must carefully evaluate each patient before using advanced devices. Adverse outcomes may discourage healthcare providers from adopting new technologies. Patients also need thorough counseling about potential risks, which adds to treatment complexity. Hospitals may face higher liability and insurance costs due to these complications. Risk management is critical in this field. Balancing innovation with patient safety remains a top priority for manufacturers, hospitals, and regulatory authorities in the neurovascular sector.
- Shortage of Skilled Professionals: The effectiveness of neurovascular devices depends heavily on trained medical professionals. Neurosurgeons and interventional radiologists require specialized skills to perform complex procedures. In many developing regions, there is a severe shortage of such experts. Even in advanced healthcare systems, high demand can strain available professionals. Limited training opportunities further restrict the adoption of advanced devices. Hospitals may have the technology but lack personnel to use it safely and effectively. This skills gap can delay treatment for patients and limit market growth. Expanding training programs and encouraging specialization are essential to overcome this challenge in the neurovascular industry.
Opportunities
- Aging Population: The global population is aging rapidly, leading to a higher prevalence of neurovascular disorders. Conditions such as stroke, aneurysms, and vascular malformations become more common with age. This trend drives the need for effective diagnostic and therapeutic devices. Hospitals and clinics are increasingly investing in neurovascular solutions to meet patient demands. Manufacturers can benefit from this growing market by offering devices that target age-related vascular issues. Early diagnosis and treatment options are also becoming a priority. As the elderly population grows, the demand for neurovascular care is expected to rise steadily, creating long-term opportunities for companies in this sector.
- Technological Advancements: Neurovascular treatments are rapidly evolving due to continuous technological innovations. Devices such as stents, thrombectomy systems, embolic coils, and flow diverters are becoming safer and more effective. These improvements expand treatment options for complex vascular conditions. Advanced devices allow for precise interventions with reduced risks. Hospitals are eager to adopt these innovations to enhance patient outcomes. Manufacturers can leverage research and development to create next-generation solutions. The ability to treat a wider range of cases with improved safety and efficacy drives adoption. Continuous innovation ensures that companies remain competitive and meet the evolving needs of healthcare providers and patients alike.
- Minimally Invasive Procedures: Minimally invasive procedures are becoming the preferred choice in neurovascular care. These procedures offer shorter recovery times, lower complication rates, and reduced hospital stays. Patients and doctors increasingly favor techniques that are less disruptive. As a result, the demand for modern neurovascular devices is growing. Tools designed for catheter-based treatments, embolization, and flow diversion are gaining traction. Healthcare providers aim to improve patient satisfaction and outcomes while minimizing risks. Manufacturers who develop user-friendly and precise devices are well-positioned to benefit. The shift toward minimally invasive solutions represents a strong opportunity to expand market share and improve healthcare delivery efficiency globally.
- Government and Healthcare Initiatives: Government programs and healthcare initiatives are supporting neurovascular care worldwide. Funding for stroke prevention, advanced diagnostics, and innovative treatment options is increasing. Investments in hospitals and medical infrastructure encourage the adoption of cutting-edge neurovascular devices. Policymakers are promoting early intervention and better management of vascular diseases. These initiatives also raise awareness among patients and healthcare providers. Companies that align with such programs can access grants, subsidies, and partnerships. Increased government and institutional support accelerates device adoption and market growth. By collaborating with healthcare authorities, manufacturers can expand their reach and contribute to improved public health outcomes in the neurovascular field.
- Emerging Markets: Emerging markets offer significant growth potential for neurovascular devices. Rapid improvements in healthcare infrastructure are expanding access to advanced treatments. Rising awareness of neurovascular disorders is prompting more patients to seek care. These regions are seeing an increase in specialized hospitals and diagnostic centers. Manufacturers can tap into new customer bases by introducing cost-effective and innovative devices. Training programs for healthcare professionals also enhance adoption rates. Growing urbanization and healthcare investments support long-term demand. By focusing on emerging markets, companies can diversify revenue streams and establish early brand recognition. These markets represent a strategic opportunity to expand global presence in the neurovascular sector.
Conclusion
In conclusion, the global neurovascular devices market is set for steady growth, driven by the rising prevalence of stroke, aneurysms, and other cerebrovascular disorders. Technological advancements, including minimally invasive devices and AI-assisted navigation, are improving treatment safety and efficiency, encouraging wider adoption in hospitals and specialized centers. Growing awareness, better healthcare infrastructure, and supportive government initiatives are further expanding access to advanced neurovascular care. Emerging markets offer additional opportunities as healthcare systems develop and patient demand rises. Overall, continued innovation, improved patient outcomes, and expanding access are expected to shape the market positively, making it a dynamic and promising sector for manufacturers and healthcare providers alike.
View More
Neurovascular Catheters Market || Neurovascular Guidewires Market || Neurostimulation Devices Market || Neuroendoscopy Devices Market || Sensorimotor Neuropathy Market || Neurorehabilitation Devices Market || Neurovascular Devices Market || Intraoperative Neurophysiological Monitoring Market || Neurophotonics Market || AI In Neurology Operating Room Market || Neurology Market || Neuroregeneration Therapy Market || Intraoperative Neuromonitoring (IONM) Market || Neurothrombectomy Devices Market || Neurofeedback Systems Market
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



