Table of Contents
Overview
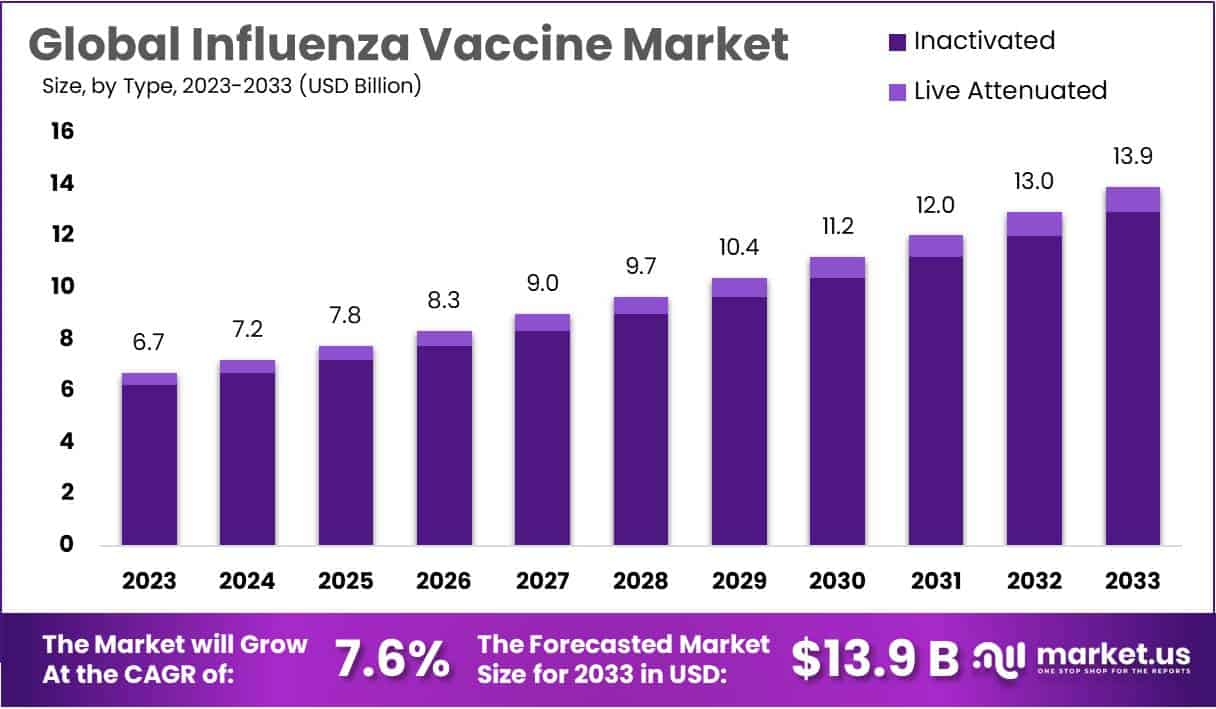
New York, NY – August 29, 2025: The global influenza vaccine market is projected to reach USD 13.9 billion by 2033, up from USD 6.7 billion in 2023, expanding at a CAGR of 7.6%. Seasonal influenza continues to impact millions globally, causing severe cases among children, the elderly, and immunocompromised patients. The persistent risk of epidemics and potential pandemics ensures steady demand. Rising incidence rates highlight the urgent need for reliable vaccines, positioning influenza immunization as a critical component of global healthcare strategies.
Government programs and reimbursement policies play a vital role in driving vaccination rates. Many countries implement national influenza immunization campaigns supported by WHO guidelines. These initiatives promote annual vaccination across all age groups, improving awareness and access. Public funding and subsidies further encourage adoption in developed and developing markets. Enhanced support from health organizations ensures higher coverage rates. Strong government involvement continues to act as a growth driver for the influenza vaccine sector worldwide.
Technological progress is reshaping vaccine development. Cell-based, recombinant, and mRNA-based influenza vaccines are enhancing safety, efficacy, and production speed. These advancements reduce reliance on egg-based manufacturing, which is less adaptable to viral mutations and time-intensive. Growing public awareness of preventive healthcare adds momentum. Campaigns highlighting influenza risks encourage annual immunization. The COVID-19 pandemic also influenced attitudes toward respiratory infections, further accelerating influenza vaccine uptake. Collectively, these factors are boosting demand and improving the competitive landscape.
Market growth is also driven by a larger high-risk population and universal vaccine research. Aging demographics and chronic conditions like diabetes and cardiovascular diseases increase influenza vulnerability. Research on universal influenza vaccines aims to provide long-lasting, broad protection against multiple strains, potentially revolutionizing the industry. Improved healthcare infrastructure, cold chain logistics, and expanded distribution networks, particularly in emerging markets, further strengthen accessibility. Regulatory bodies are streamlining approvals and offering fast-track pathways, enabling rapid rollout of innovative vaccines against evolving threats.
Key Takeaways
- The global influenza vaccine market to reach USD 13.9 billion by 2033, up from USD 6.7 billion in 2023, reflecting 7.6% CAGR.
- Inactivated influenza vaccines dominate the sector with a 92.9% share, while live attenuated vaccines grow slowly due to clinical limitations and patient restrictions.
- Quadrivalent vaccines hold the top position with 58.2% revenue share, favored for their effectiveness, affordability, and accessibility over trivalent vaccine alternatives.
- Adults represent 78.4% of the market, fueled by extensive immunization campaigns and an aging global population highly susceptible to influenza-related complications.
- The pediatric segment, though smaller, is anticipated to grow significantly as awareness and preventive healthcare measures expand for younger populations worldwide.
- Injection-based vaccines lead with 60.7% market share, while nasal spray vaccines gain momentum due to self-administration convenience and patient-friendly delivery methods.
- Hospitals and retail pharmacies capture 37.4% market share, primarily due to vaccination drives and rising influenza-related hospitalizations, with government suppliers trailing behind.
- North America remains the leading region, holding 53.9% revenue share, followed by Europe, both benefitting from robust healthcare systems and immunization programs.
- Asia-Pacific is projected to witness the fastest growth, supported by government-backed vaccination campaigns and increasing public awareness of influenza prevention.
Regional Analysis
North America held the largest revenue share of the global influenza vaccine market, accounting for 53%. The region benefits from rapid launches of effective vaccines and advanced production facilities. Strong healthcare infrastructure further supports market expansion. Rising influenza infection rates in the U.S. and Canada increase vaccine demand. Moreover, higher awareness and accessibility contribute to steady sales growth. The presence of key manufacturers in the region ensures continuous supply. Together, these factors make North America a dominant player in the market landscape.
Europe emerged as the second most significant region in the influenza vaccine market. The region’s strength lies in extended vaccination coverage targeting high-risk groups. This approach has resulted in greater immunization levels among its population. Supportive healthcare policies also encourage widespread adoption. Additionally, regulatory approvals and active distribution channels boost vaccine accessibility. European governments promote seasonal vaccination campaigns each year. Combined with the increasing elderly population, these initiatives strengthen the region’s market share. Europe is expected to maintain consistent growth in the forecast period.
Asia-Pacific is projected to register the fastest CAGR during the forecast timeline. Government initiatives play a crucial role in raising awareness about influenza vaccination. Programs to provide affordable vaccines to citizens drive adoption. Rapid population growth and urbanization further amplify vaccine demand. Countries like China, India, and Japan are focusing on expanding immunization coverage. Additionally, improvements in healthcare infrastructure support production and distribution. Local manufacturers are increasingly collaborating with global players. These combined efforts position the Asia-Pacific region as the fastest-growing influenza vaccine market globally.
Latin America and the Middle East & Africa are also witnessing significant growth prospects. Government-led vaccination campaigns are creating awareness among communities. Rising healthcare spending is improving vaccine availability in these regions. In Latin America, urban areas are showing higher adoption rates. Meanwhile, in the Middle East & Africa, international collaborations are driving access. Efforts to vaccinate vulnerable groups remain a key focus. Although growth is slower than in Asia-Pacific, both regions are expanding steadily. Their increasing contribution enhances the overall influenza vaccine market performance.
Segmentation Analysis
The global influenza vaccine market is segmented by type into inactivated and live attenuated vaccines. In 2023, inactivated vaccines held the largest market share at 92.9%. This dominance is attributed to their proven efficacy, broad adoption, and availability from multiple manufacturers. Inactivated vaccines also show strong immune responses in children aged 6–35 months. Their ability to reduce the risk of influenza A and B infections even after initial vaccination further strengthens demand. Rising prevalence of influenza supports sustained growth in this segment.
The live attenuated vaccine segment, however, is projected to grow at a slower pace. Adoption barriers and development challenges limit its expansion. These vaccines are not recommended for specific groups, such as pregnant women and individuals with asthma, which restricts usage. In contrast, inactivated vaccines remain widely prescribed. The clear preference of healthcare professionals for reliable and safer options reinforces the slower performance of live attenuated vaccines. This creates a market imbalance, with inactivated vaccines maintaining dominance globally.
Based on valency, influenza vaccines are divided into quadrivalent and trivalent types. Quadrivalent vaccines dominated in 2023, accounting for 58.2% of revenue share. This segment also showed the fastest CAGR of 7.11% during the forecast period. The key driver is their higher efficiency in protecting against viral strains. They are widely available in hospitals and clinics, ensuring accessibility. Physicians increasingly recommend quadrivalent vaccines, especially for older adults. Rising product approvals and government support further accelerate adoption, strengthening the market position of quadrivalent formulations.
Age group segmentation highlights adult dominance, with a 78.4% revenue share in 2023. This growth is linked to large vaccine doses procured by global organizations such as PAHO, UNICEF, and GAVI. Their efforts ensure wider immunization among elderly populations and reduce influenza-related hospitalizations. Pediatric vaccinations also show promising growth due to WHO-led initiatives for early-age immunization. The increasing awareness and policy support for child and adult vaccinations emphasize the importance of broad coverage. Both segments are expected to witness strong expansion over the coming years.
The market is further segmented by route of administration and distribution channels. Injectable vaccines led in 2023 with a 60.7% share, supported by the availability of intramuscular products. Nasal sprays are gaining attention due to ease of self-administration and home care suitability, making them the fastest-growing segment. Distribution is dominated by hospitals and retail pharmacies, which held 37.4% share. Government suppliers also play a vital role through extensive programs. Rising influenza cases and hospital admissions ensure steady demand, reinforcing hospitals and pharmacies as crucial vaccine providers.
Key Players Analysis
Sanofi is one of the leading companies in the influenza vaccine market. The company holds a significant share due to its strong product portfolio. Sanofi continues to expand its manufacturing facilities and geographical presence. Its global strategy focuses on meeting the rising demand for influenza vaccines across regions. With consistent investments in infrastructure, Sanofi strengthens its production capacity. This expansion helps the company maintain a competitive edge. Its leadership role reflects a strong commitment to addressing growing vaccination needs worldwide.
GlaxoSmithKline plc (GSK) is another major player in the market. GSK has built a solid reputation in vaccine development and distribution. The company operates globally, ensuring wide product availability. Its focus remains on delivering effective influenza vaccines that cater to diverse populations. By leveraging strong research and development capabilities, GSK sustains its growth trajectory. The firm also benefits from strategic partnerships and collaborations. These efforts enhance its ability to reach new markets. GSK’s contribution plays a vital role in overall market expansion.
AstraZeneca has established itself as a significant contributor to the influenza vaccine industry. The company’s focus on innovative solutions strengthens its competitive presence. AstraZeneca benefits from a strong distribution network and global outreach. Its vaccine offerings address both seasonal and pandemic influenza needs. By focusing on innovation and efficiency, the firm continues to grow its market share. The company also invests heavily in scientific research. Such initiatives help AstraZeneca meet changing healthcare demands. Its efforts align with the increasing need for effective preventive healthcare solutions.
Abbott is also recognized among the key players in this sector. The company’s vaccines are marketed worldwide, strengthening its global presence. Abbott continues to expand through facility growth and product diversification. Its focus on innovation drives strong demand for its solutions. Research and development remain central to its long-term strategy. Abbott’s efforts contribute to sustaining market competition. The firm’s ability to adapt to market needs ensures consistent growth. Together, these leading companies drive the influenza vaccine market forward with their strategic initiatives.
Challenges
- Strain Variability: One of the biggest challenges in the influenza vaccine market is strain variability. The influenza virus mutates quickly, which makes it difficult for experts to predict the dominant strains each year. When predictions are off, the effectiveness of vaccines drops. This leads to reduced protection for the population and more flu cases. Researchers must update vaccine formulations constantly, which adds pressure to manufacturers. Even with advanced surveillance systems, there is no guarantee of a perfect match. This unpredictability remains a major barrier to consistent vaccine success and public confidence.
- Short Immunity Duration: Another challenge is the short duration of immunity provided by influenza vaccines. Unlike some vaccines that protect for years, flu shots usually last for just one season. This means people need annual immunization to stay protected. Many individuals skip yearly doses, which lowers overall vaccination coverage. The short-term protection also increases the burden on healthcare systems, as campaigns must be repeated every year. For vaccine makers, this creates constant demand but also high responsibility. The recurring need for new shots is both a challenge and a strain on global immunization programs.
- Cold Chain & Storage: Maintaining an effective cold chain is a major issue for influenza vaccines. These vaccines must be stored at specific low temperatures to remain effective. Any break in the cold chain, such as during transport or storage, can damage the vaccines and make them less effective. This is especially difficult in low-resource regions where infrastructure is weak. Power shortages, poor refrigeration, and long distances to rural areas make distribution challenging. Ensuring quality and potency requires advanced logistics, which adds cost. Without reliable storage systems, many communities struggle to access safe influenza vaccines.
- Public Hesitancy: Vaccine hesitancy is another barrier in the influenza vaccine market. Many people remain skeptical due to misconceptions about safety, effectiveness, or side effects. Some believe flu shots can cause the illness, while others underestimate the risk of influenza. Limited awareness campaigns and misinformation spread through social media add to the problem. As a result, vaccination rates remain lower than required for strong community protection. Overcoming this challenge requires consistent education, transparent communication, and trust-building measures. Without addressing public hesitancy, even the most effective vaccines may not reach enough people to reduce flu outbreaks.
- High Development Costs: Developing influenza vaccines is an expensive and time-consuming process. Companies must invest heavily in research, clinical trials, and regulatory approvals. Each stage requires strict safety checks to ensure vaccines are effective. The frequent mutations of influenza viruses make the process even harder, as vaccines need regular updates. Manufacturing facilities must also follow global standards, which adds to expenses. For smaller companies, these costs can be a major barrier to entry. High development costs limit competition and slow down innovation. As a result, only a few large players dominate the influenza vaccine market.
Opportunities
- Growing Global Demand: The demand for influenza vaccines is rising worldwide. Seasonal flu continues to affect millions each year, creating a constant need for preventive measures. Increasing awareness about healthcare, particularly preventive care, is driving higher vaccine adoption rates. Public health campaigns and education initiatives are also playing an important role in boosting vaccination coverage. Additionally, the COVID-19 pandemic highlighted the importance of vaccines in protecting communities from infectious diseases. This has further accelerated trust in vaccination programs. With flu cases increasing and global populations becoming more health-conscious, demand for influenza vaccines is expected to grow steadily in the coming years.
- Technological Advancements: New vaccine technologies are creating opportunities in the influenza vaccine market. mRNA-based platforms and recombinant DNA technologies are leading the way. These advanced methods allow for faster vaccine production and improved effectiveness. Unlike traditional vaccines, they can be adapted quickly to match changing flu strains. This flexibility is a major advantage in responding to seasonal outbreaks. Moreover, biotechnology companies are investing heavily in research to create next-generation flu vaccines with longer protection. These advancements not only improve safety and efficacy but also reduce production time. As technology progresses, vaccine makers can address global health needs more efficiently.
- Government Support: Government initiatives are strengthening the influenza vaccine sector. Many countries run large-scale immunization programs to protect vulnerable populations such as children and the elderly. Public health agencies and global organizations like the WHO provide strong financial support for flu prevention. Subsidies, grants, and research funding encourage pharmaceutical companies to innovate and expand vaccine access. This backing ensures steady demand and reduces barriers for low-income communities. In addition, regulatory bodies are speeding up approvals for advanced vaccines, making them available faster. With continued government focus on disease prevention, the influenza vaccine market benefits from reliable growth opportunities worldwide.
- Expansion in Emerging Markets: Emerging economies hold untapped opportunities for influenza vaccine expansion. Countries in Asia, Africa, and Latin America are improving their healthcare systems. Rising incomes and better access to medical services mean more people can afford vaccinations. Governments in these regions are also promoting immunization programs, boosting coverage rates. Pharmaceutical companies are entering partnerships to strengthen local distribution and manufacturing. These initiatives make vaccines more affordable and accessible. As healthcare infrastructure develops further, demand for influenza vaccines will rise rapidly. With large populations and increasing health awareness, emerging markets are set to become major growth drivers for the industry.
- Universal Flu Vaccine Research: A universal flu vaccine is one of the biggest opportunities in the market. Unlike seasonal vaccines, it would provide long-term protection against multiple influenza strains. This could transform the industry by reducing the need for yearly updates. Research programs worldwide are working to make this a reality. Several biotech companies and research institutes are investing heavily in clinical trials. If successful, a universal vaccine could improve global health outcomes and reduce flu-related hospitalizations and deaths. It would also lower production costs and simplify immunization programs. This breakthrough represents a long-term growth opportunity for vaccine developers.
Conclusion
The influenza vaccine market is set for steady growth, supported by rising global demand, government programs, and continuous innovation. Strong healthcare initiatives and wider awareness campaigns are boosting vaccination coverage, especially among vulnerable groups. Advances such as cell-based and mRNA technologies are making vaccines safer, faster to produce, and more effective. While challenges like strain variability, short-term immunity, and high development costs remain, ongoing research into universal vaccines offers long-term potential. Expanding access in emerging markets also adds momentum. With growing focus on preventive healthcare, the influenza vaccine industry will continue to play a vital role in reducing seasonal flu risks and protecting public health worldwide.
View More
Influenza Diagnostics Market || Neonatal Intensive Care Respiratory Devices Market || US Respiratory Disposable Devices Market || Anesthesia and Respiratory Devices Market || Respiratory Care Devices Market || mRNA Therapeutics Market || Healthcare Cold Storage Market || Vaccine Adjuvants Market
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



