Table of Contents
Overview
New York, NY – August 29, 2025: The Contract Development and Manufacturing Organization (CDMO) Market is expanding due to rising outsourcing from pharma and biotech companies. Drug makers now depend on CDMOs to cut costs, speed up time-to-market, and access specialized expertise. This trend allows pharmaceutical firms to focus on R&D and commercialization while CDMOs manage formulation, development, and large-scale production. The growing complexity of drug development, with advanced formulations and targeted delivery systems, further strengthens the need for integrated CDMO services, including manufacturing, analytical testing, and fill-finish solutions.
The rapid expansion of biologics, biosimilars, and advanced therapies is fueling demand for CDMOs with specialized facilities and technologies. Cell and gene therapies, mRNA platforms, and high-potency APIs require unique manufacturing capabilities. Unlike small-molecule drugs, these treatments involve strict quality control and advanced processes. Patent expiries of blockbuster drugs are also boosting opportunities for generic and biosimilar production. CDMOs provide cost-efficient solutions to support companies entering these competitive markets while meeting global regulatory requirements and quality standards.
Cost efficiency and flexible manufacturing models remain key growth drivers. By outsourcing to CDMOs, pharma companies reduce heavy capital investment in infrastructure and advanced technology. Flexible and scalable manufacturing allows rapid adjustment to changing production needs, especially during fluctuating demand. Adoption of new technologies such as continuous manufacturing, automation, and single-use bioreactors is also improving efficiency. CDMOs that integrate these innovations gain a strong competitive advantage, attract long-term partnerships, and deliver high-quality solutions aligned with global industry requirements and regulatory expectations.
Regulatory expertise and industry partnerships also contribute to CDMO growth. Navigating global regulatory frameworks is challenging for drug developers, but CDMOs provide compliance support, ensuring adherence to international standards. Pharmaceutical industry consolidation through mergers and acquisitions is creating demand for long-term CDMO collaborations. Many companies now prefer partners who can manage multiple stages of the drug lifecycle. Specialized services such as sterile injectables, high-potency APIs, and niche therapies also enhance the value proposition, opening new revenue opportunities in a highly competitive market.
Geographic expansion in emerging markets is another critical growth factor. Rising healthcare investments and increasing drug demand in Asia-Pacific, Latin America, and the Middle East encourage CDMOs to establish local facilities. These expansions help lower manufacturing costs and strengthen supply chain resilience. Emerging economies are becoming attractive hubs for clinical trials, contract manufacturing, and drug distribution. By tapping into these regions, global CDMOs secure broader market access and support pharmaceutical companies in addressing the rising global demand for affordable and innovative therapies.
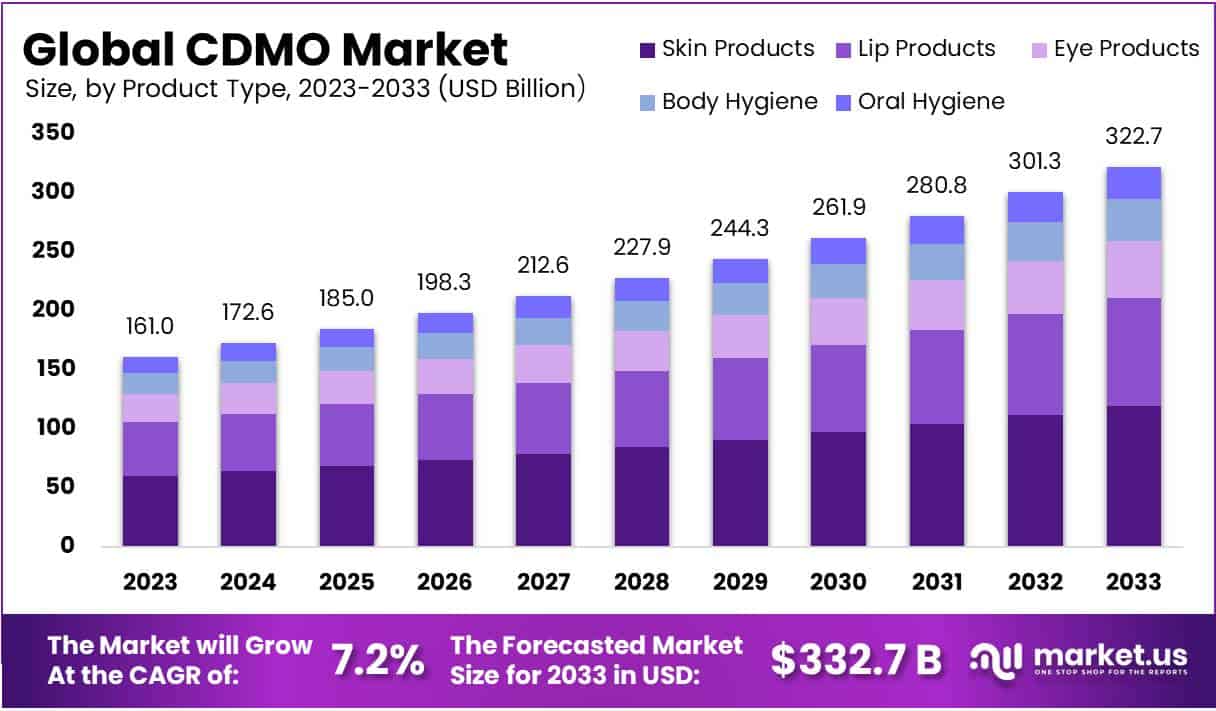
Key Takeaways
- the CDMO market to hit USD 322.7 billion by 2033, advancing steadily at a CAGR of 7.2% through 2024–2033.
- Skin products lead the market with 37.1% share in 2023, reshaping industry growth through innovation, consumer demand, and high market penetration.
- Active Pharmaceutical Ingredients (API) dominate CMO services, capturing 41.5% share, reflecting consistent demand and sustained expansion in pharmaceutical manufacturing.
- Clinical Trial Services hold a commanding 47.2% market share in 2023, strengthened by innovative partnerships and strategic advancements within drug development.
- Growing populations, Westernized lifestyles, and economic expansion are crucial forces fueling the strong upward trajectory of the global CDMO market.
- Regulatory restrictions and declining approvals for biologics and small molecules pose significant barriers to continuous CDMO market expansion globally.
- Rising partnerships with CDMOs present promising opportunities, fulfilling the increasing demand for novel therapies and expanding service portfolios across the sector.
- Increasing R&D investments, particularly within the U.S., are positively impacting CDMO market development by encouraging drug innovation and accelerated therapeutic advancements.
- North America leads with 35.1% market share in 2023, while Asia Pacific emerges as the fastest-growing region, driven by affordability and rising chronic diseases.
Regional Analysis
In 2023, North America dominated the CDMO market with a strong market share of over 35.1%, accounting for USD 56.5 billion in value. This leadership is driven by the presence of large economies like the United States and Canada. The region benefits from well-established pharmaceutical and biotechnology industries. Many global pharmaceutical giants are based in the United States, further supporting its dominance. Strong regulatory frameworks, advanced R&D facilities, and high investments in drug development strengthen North America’s position in the global CDMO market.
Asia Pacific is expected to record the fastest growth in the CDMO market during the forecast period. The region provides cost advantages compared to the United States and other developed economies, making it attractive for outsourcing. Rising healthcare demand, coupled with affordable manufacturing and research expertise, enhances its growth prospects. Pharmaceutical companies are increasingly shifting operations to countries like India and China. Growing government support, favorable policies, and expanding biopharma capabilities are fueling significant opportunities in the Asia Pacific market.
Key growth drivers in Asia Pacific also include the increasing prevalence of lifestyle-related and chronic conditions such as heart disease and diabetes. This rising disease burden is creating a strong demand for advanced therapies and clinical trials. The region also offers a large patient pool for recruitment, making it ideal for clinical studies. Moreover, the availability of skilled professionals and advanced technologies supports innovation. Together, these factors make Asia Pacific one of the most promising markets for CDMOs, with high potential for long-term growth and investment.
Segmentation Analysis
The global CDMO market is segmented by product type into hair, lip, skin, eye, body hygiene, and oral hygiene products. In 2023, skin products led the market with a 37.1% share. This dominance comes from their wide use in face creams, serums, body lotions, and sunscreen. Growing consumer focus on skincare and demand for advanced formulations will drive future growth. As a result, the skin products segment shows strong potential and will continue to be a key revenue driver in the forecast period.
Based on CMO service types, the market is divided into Active Pharmaceutical Ingredients (API), finished products, and packaging. In 2023, the API segment dominated with a 41.5% share. Growth drivers include the rising number of biologics in the pipeline and their wider adoption across therapies. The finished products segment is expected to see strong growth, fueled by increasing demand for injectables. Additionally, demand for delivery systems such as syringes and auto-injectors will further boost opportunities in this segment.
In the CRO segment, services are classified into discovery, preclinical, clinical trials, and laboratory services. In 2023, clinical trials led the market with a 47.2% share. This growth is linked to rising R&D investment, demand for outsourcing, and productive drug development. Major pharmaceutical companies are forming partnerships with CDMOs to speed up clinical trials. Patient-centric innovations and packaging improvements also support growth. Increasing partnerships for trial services and packaging solutions will strengthen the segment. Together, these factors highlight clinical trial services as a central growth driver for the CDMO market.
Key Players Analysis
Lonza from Switzerland holds the highest revenue share in the CDMO market. On the other hand, Wuxi from China is expected to record the fastest growth rate during the forecast period. Both companies are shaping industry dynamics through strong expertise and expanded service offerings. The overall market environment shows that several competitors influence pricing and quality. Smaller providers are also active, but their limited scale often restricts their long-term competitiveness in comparison with global leaders.
The CDMO industry is marked by rising competition among large players and smaller service providers. To stay competitive, many companies are shifting toward one-stop-shop solutions. These integrated services enable them to cover development, manufacturing, and regulatory support. Access to substantial financial and technical resources helps large CMOs strengthen their positions. This trend makes it harder for new entrants to compete effectively. As a result, competition among existing firms is becoming more intense, driving innovation and strategic service expansion.
The market for companion diagnostics and CDMOs is highly fragmented, with many regional and local players. Strong brand recognition, established distribution networks, and global reach give leading players a clear advantage. To improve their market rankings, pharma and biotech firms are actively pursuing strategies such as partnerships, acquisitions, and product launches. These moves help them expand their portfolios and capture larger shares of the growing outsourcing market. Such strategies are vital for sustaining long-term growth while meeting evolving client demands in the pharmaceutical and biotechnology industries.
Challenges
- High Capital Investment Requirements: CDMOs face huge costs when setting up operations. Building advanced plants, specialized labs, and quality control systems requires massive investment. Large players can afford this, but smaller firms struggle to keep up. The high barrier to entry limits competition and growth. Many small CDMOs cannot scale their infrastructure to meet client needs. This makes it hard to compete with global leaders. As a result, partnerships, mergers, or outsourcing become common survival strategies. Without enough capital, smaller firms risk losing contracts to larger competitors who can deliver at scale with efficiency.
- Regulatory Complexity: CDMOs work under strict global regulations. Agencies like the FDA, EMA, and PMDA set high compliance standards. Rules are updated frequently, making it hard for companies to adapt quickly. Meeting these requirements increases costs and creates delays. Any mistake can lead to penalties or product recalls. This not only impacts revenue but also damages reputation. Smaller CDMOs often lack the resources to manage compliance effectively. Staying up to date requires dedicated teams and constant training. The complexity of different regional rules adds another challenge. Regulatory compliance remains one of the toughest barriers to long-term growth.
- Pricing Pressure & Intense Competition: The CDMO market has many small, medium, and large players. This creates fierce competition across all service levels. To win contracts, CDMOs often lower their prices, which reduces profit margins. Larger companies with scale can manage this better, while smaller firms struggle. Constant pricing pressure makes it difficult to invest in new technologies. This slows innovation and weakens long-term competitiveness. Clients also negotiate aggressively, seeking lower costs and faster delivery. The result is a race to the bottom in pricing. For many CDMOs, maintaining quality while keeping prices competitive is a constant struggle.
- Supply Chain Vulnerabilities: CDMOs depend heavily on smooth supply chains. Any disruption creates serious challenges. Shortages in raw materials, transport delays, or trade restrictions can halt production. Geopolitical tensions and export bans make risks even higher. Such issues lead to missed deadlines and strained client relationships. Global crises, like pandemics, show how fragile supply chains can be. Smaller CDMOs are more vulnerable as they lack backup resources. Dependence on international suppliers increases the risk further. Building stronger and more resilient supply chains is costly. Yet without it, CDMOs risk losing reliability in the eyes of clients.
- Capacity Constraints: The demand for biologics, APIs, and advanced therapies is rising fast. However, manufacturing capacity often cannot keep up. Many CDMOs face project delays because they lack enough facilities or equipment. Expanding capacity requires heavy investment and time. Smaller companies struggle the most, as they cannot match the large-scale production of global leaders. This creates bottlenecks in delivery and increases costs. Clients expect fast turnaround, which puts more pressure on limited resources. Without expansion, CDMOs risk losing contracts. Meeting demand for new therapies requires better planning, more facilities, and continuous investment.
- Talent Shortage: The CDMO industry depends on skilled professionals. Roles in biologics, bioprocessing, and advanced therapies are in high demand. However, there are not enough trained experts to fill them. This shortage leads to higher salaries and rising recruitment costs. Smaller CDMOs cannot always compete with larger firms for talent. As a result, projects may slow down, and innovation is delayed. Training new staff takes time and money. The talent gap affects efficiency and limits growth potential. To stay competitive, CDMOs must invest in workforce development. Without skilled teams, delivering quality services becomes extremely difficult.
- Client Dependency Risks: Many CDMOs rely heavily on a small number of large clients. This creates dependency risks. If one major contract ends, revenue can drop sharply. The loss of a big client can also hurt market reputation. Diversifying the client base is difficult but necessary. Smaller CDMOs face this problem the most, as they cannot attract a wide range of customers. Heavy reliance also gives clients strong bargaining power. They can demand lower prices or faster timelines. CDMOs must focus on building long-term relationships with multiple clients. Spreading risk is essential for stable and sustainable growth.
Opportunities
- Growing Demand for Outsourcing: Pharma and biotech companies are relying more on outsourcing. Developing and manufacturing drugs in-house is costly and time-consuming. By outsourcing to CDMOs, companies save money, reduce risks, and speed up product launches. CDMOs also offer specialized expertise that many pharma firms do not have internally. This makes outsourcing a smart choice, especially for small and mid-sized biotech companies with limited resources. As the demand for innovative medicines grows worldwide, CDMOs that provide reliable, flexible, and cost-efficient services are well-positioned to capture this expanding market opportunity.
- Boom in Biologics and Advanced Therapies: Biologics and advanced therapies are transforming modern medicine. Demand for treatments like cell and gene therapies, biosimilars, and mRNA-based drugs is increasing rapidly. Developing and manufacturing these products requires advanced technology and strict quality standards. Many pharma companies do not have the required capabilities in-house. CDMOs that specialize in biologics, sterile manufacturing, or gene editing platforms are in high demand. This trend offers strong long-term growth potential. As healthcare systems shift towards precision medicine and advanced therapies, CDMOs that invest in these specialized areas will benefit from higher demand and long-term client partnerships.
- One-Stop-Shop Model: Pharma companies prefer working with CDMOs that offer end-to-end services. A one-stop-shop model reduces supply chain complexity and makes project management easier. CDMOs that support clients through every stage, from early drug discovery to clinical trials and large-scale manufacturing, are more attractive. This approach also strengthens customer loyalty and ensures long-term contracts. By covering multiple services under one roof, CDMOs can differentiate themselves from competitors. Clients benefit from smoother coordination, faster development timelines, and fewer risks. CDMOs adopting this integrated model are more likely to build strong partnerships and expand their market share globally.
- Expansion in Emerging Markets: Emerging regions present a big growth opportunity for CDMOs. Asia-Pacific, Latin America, and the Middle East are rapidly developing their pharmaceutical industries. Local demand for affordable medicines and international investments are fueling this growth. Establishing facilities or partnerships in these regions allows CDMOs to expand their client base and reduce operational costs. Emerging markets also provide access to skilled talent and lower production expenses. By entering these regions early, CDMOs can build strong local relationships and secure long-term contracts. This global expansion strategy will help CDMOs stay competitive and increase their presence worldwide.
- Digitalization and Smart Manufacturing: The pharmaceutical industry is moving towards digital transformation. CDMOs adopting smart manufacturing gain a clear competitive edge. Using AI, automation, and advanced analytics improves efficiency and reduces human error. Digital tools also allow better quality monitoring, faster data analysis, and shorter turnaround times. Clients benefit from reduced costs and improved product reliability. Smart manufacturing also supports regulatory compliance by ensuring traceability and real-time process control. CDMOs that invest in digital solutions can enhance productivity and meet the increasing demand for speed and accuracy in drug development. This shift is becoming a key driver of long-term growth.
- Strategic Partnerships and M&A: Partnerships and acquisitions are shaping the CDMO landscape. Large CDMOs are acquiring smaller firms to expand their expertise, geographic presence, and client networks. At the same time, partnerships with biotech startups are becoming common. These collaborations give CDMOs access to innovative products and new technologies. For smaller biotech firms, partnering with CDMOs provides resources and scalability. Mergers and acquisitions also allow CDMOs to offer broader services under one brand. This trend helps companies strengthen their global position and attract more clients. CDMOs that actively pursue strategic collaborations will remain competitive in a crowded market.
- Specialized Niche Services: Specialization is a strong opportunity for CDMOs. Areas like high-potency APIs, sterile injectables, or therapies for rare diseases require unique skills and strict compliance. Few companies can manage these specialized processes, creating a niche market for CDMOs with the right expertise. By focusing on these high-value services, CDMOs can stand out from competitors and secure premium contracts. Pharma and biotech companies often prefer trusted partners for these complex projects. Niche specialization also helps CDMOs build a reputation for quality and innovation. This strategy can lead to long-term client relationships and strong profitability in a competitive market.
Conclusion
The CDMO market is set for steady growth as pharma and biotech companies increasingly rely on outsourcing to manage complex drug development and production needs. With rising demand for biologics, advanced therapies, and cost-efficient solutions, CDMOs are becoming essential partners across the drug lifecycle. Opportunities lie in expanding services, embracing digital technologies, and strengthening global footprints, especially in emerging regions. However, challenges such as high investment needs, regulatory complexity, and talent shortages remain key barriers. Overall, CDMOs that focus on innovation, integrated solutions, and strong client relationships will be well-positioned to thrive in a competitive and evolving healthcare landscape.
View More
Topical Drugs CDMO Market || Biologics CDMO Market || Small Molecule CDMO Market || Cell and Gene Therapy CDMO Market || Biopharmaceutical CMO Market || Healthcare CMO Market || Biopharmaceutical CMO and CRO Market || Dental Implants And Prosthetics Contract Manufacturing Market || Healthcare Contract Research Outsourcing Market || Vaccine Contract Manufacturing Market || Orthopedic Contract Manufacturing Market || Diagnostic Contract Manufacturing Market
Discuss your needs with our analyst
Please share your requirements with more details so our analyst can check if they can solve your problem(s)



